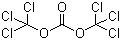
Triphosgene, chemically known as bis(halomethyl) carbonate, is an important compound in organic synthesis and is represented by the molecular formula C3Cl6O3. It is an analogue of phosgene, a toxic gas, but with enhanced stability and reduced volatility, making it a safer alternative for various applications. Triphosgene was first synthesized in the late 20th century as chemists sought to find less hazardous methods for introducing carbonyl functionalities into organic molecules.
The synthesis of triphosgene typically involves the reaction of phosgene with carbon tetrachloride, resulting in a solid compound that can be handled more safely than its gaseous counterpart. This reaction not only stabilizes the compound but also allows for easier storage and transport. The solid nature of triphosgene eliminates the risks associated with handling phosgene in gaseous form, which has been a significant concern in the chemical industry.
One of the primary applications of triphosgene is as a reagent in organic synthesis. It is widely used for the generation of isocyanates, which are crucial intermediates in the production of polyurethane foams, elastomers, and coatings. Triphosgene acts as a carbonyl source, enabling the formation of isocyanates from amines in a controlled manner. This application is particularly valuable in the production of specialty chemicals, where precise control over the reaction conditions is essential for obtaining high yields and desired product specifications.
In addition to its role in synthesizing isocyanates, triphosgene is employed in the preparation of carbonates and carbamates. These functional groups are found in a variety of pharmaceutical compounds, agrochemicals, and polymer materials. The ability of triphosgene to facilitate the formation of these groups allows chemists to develop new drug candidates and agricultural products with enhanced efficacy and reduced toxicity.
Triphosgene has also garnered attention in the field of medicinal chemistry. Researchers have utilized triphosgene in the synthesis of various bioactive compounds, particularly those that contain carbonate or carbamate functionalities. The compound's ability to introduce these functional groups in a straightforward manner makes it a valuable tool in the design and development of new therapeutic agents.
Moreover, triphosgene's utility extends to the field of polymer chemistry, where it is used to modify existing polymers or to create new polymeric materials with tailored properties. By incorporating carbonate or carbamate moieties into polymer chains, chemists can enhance the thermal stability, mechanical strength, and chemical resistance of the resulting materials. This versatility is particularly important in developing high-performance polymers for use in demanding applications such as aerospace, automotive, and electronics.
In summary, triphosgene is a significant compound in organic synthesis, providing a safer alternative to phosgene for the generation of isocyanates, carbonates, and carbamates. Its applications span various fields, including pharmaceuticals, agrochemicals, and materials science, making it an invaluable reagent for chemists. The continued exploration of triphosgene's properties and potential applications may lead to further advancements in organic synthesis and material development.
References
2024. Pyridyl-Substituted Ureas and Carbamates: Synthesis and Application (A Review). Russian Journal of General Chemistry, 94(14).
DOI: 10.1134/s1070363224140093
2024. Nafithromycin (MIQNAF®): ultramodern lactone ketolide designed to treat community acquired bacterial pneumonia (CABP). Medicinal Chemistry Research, 33(8).
DOI: 10.1007/s00044-024-03281-5
2023. Design and development of an efficient convergent synthetic strategy for novel β-lactam enhancer zidebactam (WCK 5107). Chemical Papers, 78(3).
DOI: 10.1007/s11696-023-03176-6
|


















































 GHS05;GHS06 Danger Details
GHS05;GHS06 Danger Details