Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Mascot I.E. CO.,Ltd. | China | |||
|---|---|---|---|---|
 | www.mascotchem.com | |||
 | +86 (519) 8501-0339 +86 13584504415 | |||
 | +86 (519) 8501-0366 | |||
 | info@mascotchem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2006 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Institute of Petrochemistry, Heilongjiang Academy of Science | China | |||
|---|---|---|---|---|
 | www.oledchem.cn | |||
 | +86 (451) 8262-3696 +86 13945093324 | |||
 | +86 (451) 8262-3696 | |||
 | oledchem@gmail.com lkiwi2000@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1962 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Boron Molecular Inc. | USA | |||
|---|---|---|---|---|
 | www.boronmolecular.com | |||
 | +1 (919) 981-4441 | |||
 | +1 (919) 981-4442 | |||
 | sales@boronmolecular-usa.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Chemoki Synthesi-Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemoki.com | |||
 | +86 (21) 5108-6038 | |||
 | +86 (21) 5186-1608 | |||
 | chemoki.offer@gmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Porphyrin Systems | Germany | |||
|---|---|---|---|---|
 | www.porphyrin-systems.de | |||
 | +49 (40) 6466-5860 | |||
 | +49 (40) 6466-5861 | |||
 | hombrecher@porphyrin-systems.de | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Hohance Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hohance.com | |||
 | +86 (21) 3111-5312 | |||
 | +86 (21) 3111-5317 | |||
 | info@hohance.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Boroncore LLC | China | |||
|---|---|---|---|---|
 | www.boroncore.com | |||
 | +86 (571) 8558-6753 | |||
 | custom@boroncore.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Valiant Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.valiant-cn.com | |||
 | +86 (535) 610-1878 | |||
 | +86 (535) 638-6989 | |||
 | jiangyanan@valiant-cn.com chenxiaowei@valiant-cn.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Pure Chemistry Scientific Inc | USA | |||
|---|---|---|---|---|
 | www.purechems.com | |||
 | +1 (888) 588-9418 | |||
 | +1 (617) 206-9595 | |||
 | sales@purechems.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Yantai Jiacheng Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ytjcchem.com | |||
 | +86 (535) 630-3697 +86 18660586989 | |||
 | +86 (535) 630-3697 | |||
 | sales@ytjcchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Yantai Xianhua Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xianhualc.com | |||
 | +86 (535) 693-4138 +86 18663898285 | |||
 | +86 (535) 693-4137 | |||
 | sales@xianhualc.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Dalian Bangyong Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dl-bykj.cn | |||
 | +86 (411) 6685-8498 | |||
 | sales@dl-bykj.cn | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Frontier Specialty Chemicals | USA | |||
|---|---|---|---|---|
 | www.frontierspecialtychemicals.com | |||
 | +1 (435) 753-1901 | |||
 | +1 (435) 753-6731 | |||
 | sales@frontierspecialtychemicals.com | |||
| Chemical manufacturer since 1975 | ||||
| Syntech Labs, Inc. | USA | |||
|---|---|---|---|---|
 | www.syntechlabs.com | |||
 | +1 (732) 545-8380 | |||
 | +1 (732) 545-6881 | |||
 | info@syntechlabs.com | |||
| Chemical manufacturer | ||||
| Archimica Inc. | USA | |||
|---|---|---|---|---|
 | www.archimica.com | |||
 | +1 (302) 994-3043 | |||
 | +1 (302) 994-3475 | |||
 | contact@archimica.com | |||
| Chemical manufacturer since 1946 | ||||
| AOBChem USA | USA | |||
|---|---|---|---|---|
 | www.aobchem.com | |||
 | +1 (323) 382-7678 | |||
 | +1 (213) 687-8698 | |||
 | sales@aobchem.com | |||
| Chemical manufacturer | ||||
| Beijing Bayi Space LCD Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bayi.com.cn | |||
 | +86 (10) 6976-5588 ex 8068 +86 18600516188 | |||
 | +86 (10) 6976-0560 | |||
 | zhangshupo@aliyun.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2004 | ||||
| Trigona oHG | Germany | |||
|---|---|---|---|---|
 | www.trigona.de | |||
 | +49 (611) 962-5283 | |||
 | +49 (611) 962-9032 | |||
 | info@trigona.de | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Alfa Aesar. | USA | |||
|---|---|---|---|---|
 | www.alfa.com | |||
 | +1 (978) 521-6300 | |||
 | +1 (978) 521-6350 | |||
 | info@alfa.com | |||
| Chemical manufacturer | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Acros Organics | Belgium | |||
|---|---|---|---|---|
 | www.acros.com | |||
 | +86 (21) 5258-1100 | |||
 | +86 (21) 5258-0119 | |||
 | info@acros.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Boric acid |
|---|---|
| Name | 2-Naphthaleneboronic acid |
| Synonyms | 2-Naphthylboronic acid |
| Molecular Structure | 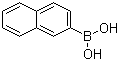 |
| Molecular Formula | C10H9BO2 |
| Molecular Weight | 171.99 |
| CAS Registry Number | 32316-92-0 |
| EC Number | 628-070-3 |
| SMILES | B(C1=CC2=CC=CC=C2C=C1)(O)O |
| Density | 1.2±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 269-275 °C (Expl.) |
| Index of Refraction | 1.639, Calc.* |
| Boiling Point | 381.9±25.0 °C (760 mmHg), Calc.* |
| Flash Point | 184.8±23.2 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
2-Naphthaleneboronic acid is a chemical compound that belongs to the family of boronic acids, which are widely used in organic synthesis and medicinal chemistry. It consists of a naphthalene ring attached to a boronic acid group (-B(OH)2) at the second position of the aromatic ring. The compound is of significant interest due to its versatile reactivity, particularly in the formation of boronic esters and its utility in various catalytic reactions. The discovery of 2-naphthaleneboronic acid is part of a broader field of research into boronic acids, which gained prominence in the 20th century due to their ability to form stable complexes with diols and other ligands. Boronic acids, including 2-naphthaleneboronic acid, have become essential tools in organic chemistry, particularly for their use in Suzuki-Miyaura coupling reactions. This reaction allows for the efficient formation of carbon-carbon bonds, making it a cornerstone of modern organic synthesis. The development and application of boronic acids, including 2-naphthaleneboronic acid, have been pivotal in the synthesis of pharmaceuticals, agrochemicals, and materials science. The primary application of 2-naphthaleneboronic acid is in Suzuki-Miyaura coupling reactions, where it acts as a key reagent for the formation of biaryl compounds. This reaction involves the coupling of an organoboron compound with an organic halide in the presence of a palladium catalyst, and it has been widely used in the synthesis of complex organic molecules. The ability of 2-naphthaleneboronic acid to participate in such reactions is highly valuable in the production of pharmaceuticals, where it can be used to create molecules with significant biological activity. For example, the synthesis of complex molecules used in cancer therapy, anti-inflammatory drugs, and other therapeutic agents often involves Suzuki-Miyaura coupling reactions with boronic acids like 2-naphthaleneboronic acid. Beyond its role in Suzuki-Miyaura reactions, 2-naphthaleneboronic acid has also been utilized in the preparation of various organic materials. For instance, it has been incorporated into the synthesis of conjugated materials, which are of interest in organic electronics, such as organic light-emitting diodes (OLEDs) and organic solar cells. The electronic properties of naphthalene derivatives, combined with the versatility of boronic acids, make 2-naphthaleneboronic acid a useful building block in the design of advanced materials with potential applications in optoelectronics. Additionally, 2-naphthaleneboronic acid has been studied for its interactions with biological systems. In particular, boronic acids are known to form reversible covalent bonds with diols, a property that has led to their exploration as potential agents for sensing and drug delivery. In this context, 2-naphthaleneboronic acid could be used in the design of biosensors or in the development of therapeutics that target specific biomolecules, such as carbohydrates or proteins with cis-diol groups. Its ability to bind to specific targets in biological systems makes it a valuable compound in chemical biology and medicinal chemistry. In summary, 2-naphthaleneboronic acid is a boronic acid derivative with important applications in organic synthesis, particularly in Suzuki-Miyaura coupling reactions for the construction of biaryl compounds. Its role in the development of pharmaceuticals, materials science, and potential biosensing applications underscores its significance in both academic and industrial chemistry. Through its versatile reactivity and utility in diverse chemical processes, 2-naphthaleneboronic acid continues to be a key compound in the advancement of synthetic and medicinal chemistry. References 2009. Carbonic anhydrase inhibitors. Inhibition of the fungal beta-carbonic anhydrases from Candida albicans and Cryptococcus neoformans with boronic acids. Bioorganic & Medicinal Chemistry Letters, 19(10). DOI: 10.1016/j.bmcl.2009.03.147 2021. Dual Roles of TBHP-Enabled Regioselective Hydroetherification of (Trifluoromethyl)alkenes with Boronic Acids: Access to α-Trifluoromethyl β-Aryloxy Tertiary Alcohols. Synthesis, 53(19). DOI: 10.1055/a-1481-2023 2024. Reactive extraction for the separation of glyceric acid from aqueous solutions with 2-naphthaleneboronic acid and tri-octyl methyl ammonium chloride. Biotechnology and Bioprocess Engineering, 29(3). DOI: 10.1007/s12257-024-00110-9 |
| Market Analysis Reports |