Online Database of Chemicals from Around the World
| Wuhan Demeikai Biotechnology Co., Ltd | China | |||
|---|---|---|---|---|
 | www.dxhgnb.com | |||
 | +86 +86 (027) 5045-8986 +86 17192727886 | |||
 | sare@dmksw.xin wzh@dmksw.xin | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Send Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shsendpharma.com | |||
 | +86 (0)21) 5808-8081 | |||
 | sale@shsendpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Quinoline compound |
|---|---|
| Name | 6-(Bis-(2,2,2-trifluoroethyl)amino)-4-trifluoromethyl-1H-quinolin-2-one |
| Synonyms | LGD-2226 |
| Molecular Structure | 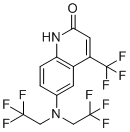 |
| Molecular Formula | C14H9F9N2O |
| Molecular Weight | 392.22 |
| CAS Registry Number | 328947-93-9 |
| EC Number | 803-690-9 |
| SMILES | C1=CC2=C(C=C1N(CC(F)(F)F)CC(F)(F)F)C(=CC(=O)N2)C(F)(F)F |
| Solubility | 0.7661 mg/L (25 °C water) |
|---|---|
| Density | 1.5±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.463, Calc.* |
| Melting point | 152.96 °C |
| Boiling Point | 377.15 °C, 364.4±42.0 °C (760 mmHg), Calc.* |
| Flash Point | 174.2±27.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS08 Warning Details GHS08 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H373 Details | ||||||||
| Safety Statements | P260-P319-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
6-(Bis-(2,2,2-trifluoroethyl)amino)-4-trifluoromethyl-1H-quinolin-2-one is a notable compound in the field of medicinal chemistry, primarily due to its unique structure and potential biological activities. This quinoline derivative features multiple trifluoromethyl groups and a bis(trifluoroethyl) amino substituent, contributing to its chemical stability and lipophilicity, which are important for drug-like properties. The discovery of this compound can be traced back to the growing interest in fluorinated organic compounds, particularly in the development of pharmaceuticals. Fluorine is known to enhance the metabolic stability and bioavailability of drugs, making fluorinated compounds an area of active research. The presence of the quinoline core in this structure adds to its significance, as quinolines are recognized for their diverse pharmacological profiles, including antimalarial, anticancer, and antimicrobial activities. The synthesis of 6-(bis-(2,2,2-trifluoroethyl)amino)-4-trifluoromethyl-1H-quinolin-2-one typically involves several key steps. Initially, a suitable quinoline precursor is functionalized through various reactions, such as nucleophilic substitution or coupling reactions, to introduce the trifluoromethyl and bis(trifluoroethyl) amino groups. These synthetic routes are often optimized to enhance yield and purity, enabling researchers to obtain the target compound efficiently. In terms of applications, the unique chemical structure of 6-(bis-(2,2,2-trifluoroethyl)amino)-4-trifluoromethyl-1H-quinolin-2-one positions it as a candidate for drug development. The trifluoromethyl groups can influence the compound's interaction with biological targets, potentially enhancing its efficacy and selectivity. Preliminary studies have indicated that compounds with similar structural motifs exhibit promising biological activities, making this compound a valuable addition to the medicinal chemistry toolbox. Moreover, the compound’s potential as a building block for further chemical modifications is significant. The trifluoromethyl and bis(trifluoroethyl) amino groups can participate in various chemical reactions, allowing for the synthesis of a wide range of derivatives. This versatility is advantageous in the search for new therapeutic agents, as it enables the exploration of different substituents that may improve pharmacological properties. Research into the biological activities of 6-(bis-(2,2,2-trifluoroethyl)amino)-4-trifluoromethyl-1H-quinolin-2-one is ongoing, with scientists investigating its potential effects on various biological pathways. The compound’s lipophilic nature may facilitate its penetration into biological membranes, enhancing its activity against targeted cells. As the field of medicinal chemistry continues to evolve, compounds like this one represent important milestones in the quest for novel therapeutics. In summary, 6-(bis-(2,2,2-trifluoroethyl)amino)-4-trifluoromethyl-1H-quinolin-2-one is a fluorinated quinoline derivative with significant potential in medicinal chemistry. Its unique structural features and the presence of trifluoromethyl groups may enhance its biological activity and pharmacokinetic properties. Ongoing research will likely uncover further applications and lead to the development of new compounds based on this promising structure. References 2007. Discovery of an androgen receptor modulator pharmacophore based on 2-quinolinones. Bioorganic & Medicinal Chemistry Letters, 17(6). DOI: 10.1016/j.bmcl.2007.01.007 2007. Novel selective androgen receptor modulators: SAR studies on 6-bisalkylamino-2-quinolinones. Bioorganic & Medicinal Chemistry Letters, 17(6). DOI: 10.1016/j.bmcl.2007.01.001 2007. Substituted 6-(1-Pyrrolidine)quinolin-2(1H)-ones as Novel Selective Androgen Receptor Modulators. Journal of Medicinal Chemistry, 50(20). DOI: 10.1021/jm070231h" |
| Market Analysis Reports |