Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Fuxin Zangyou Chemical Research and Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zyflu.com | |||
 | +86 (418) 226-1222 | |||
 | +86 (418) 653-0589 | |||
 | sales@zyflu.com zangyou@sohu.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Lee Biosolutions, Inc. | USA | |||
|---|---|---|---|---|
 | www.leebio.com | |||
 | +1 (314) 968-1091 | |||
 | +1 (314) 968-9851 | |||
 | info@leebio.com | |||
| Chemical manufacturer | ||||
| Melford Laboratories Limited | UK | |||
|---|---|---|---|---|
 | www.melford.co.uk | |||
 | +44 (1449) 741-178 | |||
 | +44 (1449) 741-217 | |||
 | pfranklin@melford.co.uk sales@melford.co.uk | |||
| Chemical manufacturer since 1985 | ||||
| Ricci Chimica | Italy | |||
|---|---|---|---|---|
 | www.riccichimica.com | |||
 | +39 (75) 692-9241 | |||
 | +39 (75) 592-6595 | |||
 | info@riccichimica.com | |||
| Chemical manufacturer | ||||
| AOBChem USA | USA | |||
|---|---|---|---|---|
 | www.aobchem.com | |||
 | +1 (323) 382-7678 | |||
 | +1 (213) 687-8698 | |||
 | sales@aobchem.com | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic acid |
|---|---|
| Name | 4-(Trifluoromethoxy)benzoic acid |
| Molecular Structure | 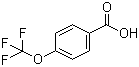 |
| Molecular Formula | C8H5F3O3 |
| Molecular Weight | 206.12 |
| CAS Registry Number | 330-12-1 |
| EC Number | 206-352-3 |
| SMILES | C1=CC(=CC=C1C(=O)O)OC(F)(F)F |
| Density | Predicted data is generated using the ACD/Labs Percepta Platform - PhysChem Module, Calc.* |
|---|---|
| Melting point | 151-154 °C (Expl.) |
| Index of Refraction | Flash point93.0±25.9 °C, Calc.* |
| Boiling Point | Density1.4±0.1 g/cm³, Calc.* |
| Flash Point | Enthalpy of vaporization49.3±3.0 kJ/mol, Calc.* |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
4-(Trifluoromethoxy)benzoic acid is a chemical compound that is primarily utilized in the field of organic chemistry and material science. The substance consists of a benzoic acid structure with a trifluoromethoxy group (-OCF₃) attached at the para position relative to the carboxyl group (-COOH). The trifluoromethoxy group, which is an electronegative functional group, significantly influences the chemical properties of the compound, such as its stability and reactivity. The discovery and development of 4-(Trifluoromethoxy)benzoic acid can be traced back to the growing interest in trifluoromethylated compounds, particularly in the context of fine chemicals and pharmaceutical synthesis. The trifluoromethoxy group imparts unique characteristics to organic molecules, such as increased lipophilicity, metabolic stability, and improved binding affinity in certain biochemical environments. These attributes make trifluoromethoxybenzoic acid derivatives valuable in various applications, especially in the design of drug molecules and other bioactive substances. In terms of applications, 4-(Trifluoromethoxy)benzoic acid is used as a building block in the synthesis of a wide range of compounds, including pharmaceuticals, agrochemicals, and materials science products. The introduction of a trifluoromethoxy group to the benzoic acid structure often enhances the compound's performance in certain biological and chemical processes. In particular, the compound and its derivatives have been studied for their potential in developing anti-inflammatory agents and other therapeutics, due to their ability to interact with key enzymes and biological receptors. This compound is also employed in the preparation of polymer materials. Trifluoromethoxy-substituted aromatic compounds are known to have high thermal stability and resistance to oxidative degradation, making them suitable for high-performance coatings, films, and other materials. The incorporation of 4-(Trifluoromethoxy)benzoic acid into polymer matrices can improve the material's properties, such as its mechanical strength, chemical resistance, and thermal stability. Moreover, 4-(Trifluoromethoxy)benzoic acid can be used in synthetic chemistry as a reagent or intermediate for more complex transformations. It is involved in reactions that require the trifluoromethoxy group as a functional handle, such as nucleophilic substitution and cross-coupling reactions. These reactions are useful in the synthesis of a variety of organic compounds, including other fluorinated benzoic acids and complex pharmaceuticals. In summary, 4-(Trifluoromethoxy)benzoic acid is an important chemical intermediate in the synthesis of pharmaceuticals, agrochemicals, and high-performance materials. Its unique trifluoromethoxy group makes it valuable for enhancing the stability, reactivity, and bioactivity of organic molecules. Its applications in drug development, materials science, and synthetic chemistry highlight its utility in modern chemical and industrial processes. References 1993. QSAR's from similarity matrices. Technique validation and application in the comparison of different similarity evaluation methods. Journal of Medicinal Chemistry, 36(20). DOI: 10.1021/jm00072a012 2016. Screening of efficient siRNA carriers in a library of surface-engineered dendrimers. Scientific Reports, 6, 25069. DOI: 10.1038/srep25069 2022. Selective Oxidations Using a Cytochrome P450 Enzyme Variant Driven with Surrogate Oxygen Donors and Light. Chemistry, 28(40). DOI: 10.1002/chem.202201366 |
| Market Analysis Reports |