Online Database of Chemicals from Around the World
| Hangzhou Chuan Shuo Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.transol.cn | |||
 | +86 (571) 8760-9021 | |||
 | qian@transol.cn | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Amine |
|---|---|
| Name | 2-Methoxymethyl-p-phenylenediamine |
| Synonyms | 2-(methoxymethyl)benzene-1,4-diamine |
| Molecular Structure | 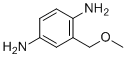 |
| Molecular Formula | C8H12N2O |
| Molecular Weight | 152.19 |
| CAS Registry Number | 337906-36-2 |
| EC Number | 679-526-3 |
| SMILES | COCC1=C(C=CC(=C1)N)N |
| Density | 1.1±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 296.0±25.0 °C 760 mmHg (Calc.)* |
| Flash point | 150.2±16.8 °C (Calc.)* |
| Index of refraction | 1.608 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS06;GHS07;GHS09 Danger Details GHS06;GHS07;GHS09 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301+H311+H331-H301-H311-H317-H319-H332-H411 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P262-P264-P264+P265-P270-P271-P272-P273-P280-P301+P316-P302+P352-P304+P340-P305+P351+P338-P316-P317-P321-P330-P333+P317-P337+P317-P361+P364-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
2-Methoxymethyl-p-phenylenediamine is an aromatic amine compound belonging to the class of substituted phenylenediamines, which are widely used in industrial and cosmetic applications, particularly as hair dye intermediates. Its chemical structure features a benzene ring substituted with amino groups at the para (1,4) positions, and a methoxymethyl (-CH2OCH3) group at the 2-position. The IUPAC name is 4-amino-2-(methoxymethyl)aniline. This compound was developed as part of the search for effective oxidative hair dye precursors that provide desirable shades while minimizing skin sensitization and toxicity. It is typically synthesized via multi-step reactions involving selective substitution on a phenylenediamine core. The introduction of the methoxymethyl substituent influences the electron distribution of the aromatic ring and modifies the reactivity of the amino groups, thereby affecting the oxidative color formation process in dye formulations. 2-Methoxymethyl-p-phenylenediamine is used mainly in oxidative hair coloring systems, where it acts as a primary intermediate. In such systems, it undergoes oxidation (usually with hydrogen peroxide) in the presence of couplers to produce colored molecules that deposit in the hair shaft. The methoxymethyl group can affect the shade intensity, fastness, and tone of the resulting dye, often contributing to natural brown or black hues depending on formulation conditions and the presence of other ingredients. From a chemical perspective, the compound is a crystalline solid, usually off-white to pale beige in color, with limited solubility in water and greater solubility in alcohols and glycol solvents. The amino groups at the 1- and 4-positions make it a strong nucleophile and susceptible to oxidation, which is essential for its role in color formation. The methoxymethyl group adds steric hindrance and changes the overall polarity of the molecule. In terms of safety, as with many substituted phenylenediamines, 2-methoxymethyl-p-phenylenediamine has been evaluated for potential skin sensitization and toxicological effects. Regulatory bodies in the European Union and other regions have conducted safety assessments of this compound as part of broader hair dye regulation. Based on such evaluations, its use in cosmetic products is generally permitted within specific concentration limits and with mandatory safety labeling. It is not intended for use in products applied to eyelashes or eyebrows, due to the risk of severe eye injury. Analytical detection and quality control of 2-methoxymethyl-p-phenylenediamine in formulations are typically performed using high-performance liquid chromatography (HPLC), often coupled with UV detection or mass spectrometry. These methods allow for monitoring of purity, degradation products, and compliance with regulatory standards. Spectroscopic techniques such as nuclear magnetic resonance (NMR) and infrared (IR) spectroscopy are used to confirm molecular identity and structural features during synthesis and formulation development. Environmentally, the compound is considered a pollutant of concern in the context of cosmetic wastewater due to its potential toxicity to aquatic organisms. Proper disposal and wastewater treatment practices are necessary to mitigate its impact on ecosystems. In summary, 2-methoxymethyl-p-phenylenediamine is a specialized aromatic amine used primarily in oxidative hair dye formulations. Its chemical structure enables it to participate effectively in color-generating reactions, while the methoxymethyl substituent fine-tunes its reactivity and shade characteristics. Regulatory oversight ensures that its use is controlled for safety, and it continues to be a relevant compound in the formulation of modern hair color products. References 2024. Dermale und aerogene Berufsstoff-Exposition im Friseurhandwerk. hautnah. DOI: 10.1007/s12326-024-00672-8 2019. 2-Methoxymethyl-para-phenylenediamine-containing hair dye as a less allergenic alternative for para-phenylenediamine-allergic individuals. Journal of the American Academy of Dermatology, 80(3). DOI: 10.1016/j.jaad.2018.06.019 2016. Continuous usage of a hair dye product containing 2-methoxymethyl-para-phenylenediamine by hair-dye-allergic individuals. The British journal of dermatology, 174(5). DOI: 10.1111/bjd.14390 |
| Market Analysis Reports |