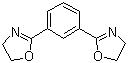
1,3-Bis(4,5-dihydro-2-oxazolyl)benzene, a compound with the molecular formula C13H12N2O2, is a member of the oxazole family known for its unique bicyclic structure. This compound was first synthesized in the mid-20th century as part of research efforts to develop heterocyclic compounds with potential applications in various fields, including medicinal chemistry and material science.
The synthesis of 1,3-bis(4,5-dihydro-2-oxazolyl)benzene typically involves the reaction of substituted benzene derivatives with oxazoles, resulting in the formation of this bis-oxazole compound. The resulting structure features two oxazole rings connected to a benzene core, which contributes to its interesting chemical and physical properties.
One of the prominent applications of 1,3-bis(4,5-dihydro-2-oxazolyl)benzene is in the field of medicinal chemistry. Compounds containing oxazole rings have been studied for their biological activity, including antimicrobial, antifungal, and anti-inflammatory properties. Preliminary research has suggested that this particular compound may exhibit potential as a therapeutic agent, although further studies are needed to explore its pharmacological effects and mechanisms of action.
In addition to its medicinal applications, 1,3-bis(4,5-dihydro-2-oxazolyl)benzene has shown promise in the development of novel materials. Its unique structure allows for the incorporation of functional groups that can enhance the thermal and mechanical properties of polymers. This makes it a candidate for use in high-performance materials, particularly in applications requiring durability and resistance to environmental degradation.
Moreover, research has indicated that this compound could serve as a building block in the synthesis of advanced materials, such as organic semiconductors and dyes. The ability to modify the oxazole rings provides opportunities for tailoring the electronic properties of materials, making them suitable for applications in organic electronics and optoelectronics.
Another area of interest for 1,3-bis(4,5-dihydro-2-oxazolyl)benzene is in the field of chemical sensors. Due to its structural characteristics, it has the potential to be used in the development of sensors capable of detecting specific analytes, including biomolecules and environmental pollutants. The incorporation of this compound into sensor devices may enhance sensitivity and selectivity, providing valuable tools for analytical applications.
Despite its potential, research on 1,3-bis(4,5-dihydro-2-oxazolyl)benzene is still in the early stages, and further exploration of its properties and applications is needed. As studies continue to uncover the versatility of this compound, it may pave the way for innovative solutions in medicine, materials science, and analytical chemistry.
In summary, 1,3-bis(4,5-dihydro-2-oxazolyl)benzene is a compound with significant potential in various fields, particularly in medicinal chemistry and material development. Its unique oxazole structure and the ability to modify its properties open up new avenues for research and application, making it a valuable compound for future exploration.
References
2022. Synthesis of 2-Oxazolines and Oxazoles. Science of Synthesis.
URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-115-00863
|



























 GHS07 Warning Details
GHS07 Warning Details