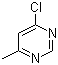
6-Chloro-4-methylpyrimidine is a heterocyclic compound containing both a chloro group and a methyl group attached to a pyrimidine ring. Pyrimidine itself is a six-membered aromatic ring that contains two nitrogen atoms at positions 1 and 3. The presence of the chloro and methyl substituents on the ring modifies the compound’s chemical properties, influencing its reactivity and utility in various chemical applications.
The structure of 6-chloro-4-methylpyrimidine features a chlorine atom at the 6-position and a methyl group at the 4-position of the pyrimidine ring. This substitution pattern is particularly important because the chloro group is an electron-withdrawing group, while the methyl group is an electron-donating group. As a result, the combination of these two substituents can influence the electron density of the pyrimidine ring, affecting the compound’s behavior in chemical reactions.
6-Chloro-4-methylpyrimidine is commonly used as an intermediate in organic synthesis, especially in the preparation of more complex molecules for use in pharmaceuticals, agrochemicals, and materials science. The electron-withdrawing nature of the chloro group and the electron-donating effect of the methyl group make this compound useful for various transformations in synthetic chemistry.
One of the primary applications of 6-chloro-4-methylpyrimidine is in the synthesis of biologically active molecules. Pyrimidine derivatives are widely studied in medicinal chemistry due to their role in many biological processes. Compounds with pyrimidine rings are found in a variety of pharmaceutical agents, such as antiviral, anticancer, and anti-inflammatory drugs. The functional groups on the pyrimidine ring, such as the chloro and methyl groups, can be strategically modified to enhance the pharmacological properties of the resulting compounds.
For example, the chloro group on 6-chloro-4-methylpyrimidine can be replaced or reacted with other nucleophiles in substitution reactions, allowing for the creation of novel compounds with altered biological activity. The methyl group, on the other hand, may serve to increase the lipophilicity of the molecule, improving its membrane permeability or affecting its distribution in the body. These modifications are key in the design of new drug candidates.
The reactivity of 6-chloro-4-methylpyrimidine also makes it a useful building block for synthesizing more complex heterocyclic compounds. Pyrimidines are often involved in ring closure reactions, where the positions of the chloro and methyl groups can guide the formation of new rings, leading to the creation of tricyclic or polycyclic compounds. These types of molecules are often seen in drug discovery and materials science for their unique chemical properties and stability.
Additionally, 6-chloro-4-methylpyrimidine can be used in the synthesis of agrochemicals. Pyrimidine derivatives are employed in the development of herbicides, fungicides, and insecticides due to their ability to interact with biological systems in specific ways. The ability to modify the functional groups on the pyrimidine ring allows chemists to fine-tune the activity of these compounds for targeted pest control.
The versatility of 6-chloro-4-methylpyrimidine in organic synthesis makes it a valuable intermediate for the development of a wide range of chemical products. By selectively modifying the chloro and methyl substituents, it is possible to create molecules with diverse properties, including improved stability, solubility, and reactivity.
In summary, 6-chloro-4-methylpyrimidine is an important heterocyclic compound with applications in pharmaceutical and agrochemical synthesis. The combination of chloro and methyl groups on the pyrimidine ring influences its reactivity and provides opportunities for further functionalization, making it a key building block in the development of new drugs, agrochemicals, and materials. The unique properties of this compound make it a valuable tool in synthetic chemistry, contributing to the creation of novel and functional molecules with specific biological and chemical activities.
|
























 GHS07 Warning Details
GHS07 Warning Details