Online Database of Chemicals from Around the World
| Alfa Chemistry | USA | |||
|---|---|---|---|---|
 | www.alfa-chemistry.com | |||
 | +1 (201) 478-8534 | |||
 | +1 (516) 927-0118 | |||
 | inquiry@alfa-chemistry.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Fenhe Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fenhechem.com | |||
 | +86 (021) 3392-6068 | |||
 | julius.wei@fenhechem.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Flavors and spices >> Synthetic spice >> Aldehyde fragrance >> Acyclic aliphatic aldehyde |
|---|---|
| Name | Sodium nitromalonaldehyde |
| Synonyms | Sodium 2-nitro-1,3-dioxopropan-2-ide |
| Molecular Structure | 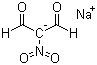 |
| Molecular Formula | C3H2NNaO4 |
| Molecular Weight | 139.04 |
| CAS Registry Number | 34461-00-2 (126901-44-8) |
| EC Number | 636-459-4 |
| SMILES | C(=C(C=O)[N+](=O)[O-])[O-].[Na+] |
| Melting point | 120-124 °C |
|---|---|
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H314-H332 Details | ||||||||||||||||||||
| Safety Statements | P260-P261-P264-P270-P271-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P330-P362+P364-P363-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Sodium nitromalonaldehyde is a fascinating chemical compound known for its reactive nature and importance in organic synthesis. This compound, characterized by a nitro group attached to a malonaldehyde backbone, plays a pivotal role in various chemical reactions and has applications in both research and industrial processes. The discovery of sodium nitromalonaldehyde can be traced to studies on reactive aldehydes and nitro compounds. Malonaldehyde, or propanedial, is a simple dialdehyde known for its reactivity, particularly in polymerization and condensation reactions. By introducing a nitro group into the malonaldehyde structure, chemists created sodium nitromalonaldehyde, a compound with enhanced reactivity that opens up new possibilities for chemical synthesis. One of the key applications of sodium nitromalonaldehyde is in organic synthesis, where it serves as a versatile intermediate in the construction of more complex molecules. The nitro group in the compound is electron-withdrawing, making the adjacent carbon atoms highly reactive and suitable for nucleophilic addition reactions. This property allows sodium nitromalonaldehyde to participate in various chemical transformations, including the synthesis of heterocyclic compounds, which are vital in the development of pharmaceuticals, agrochemicals, and dyes. In the pharmaceutical industry, sodium nitromalonaldehyde is used as a building block in the synthesis of biologically active molecules. Its reactivity facilitates the formation of key intermediates in the production of drugs, particularly those that require the incorporation of nitro or aldehyde functionalities. The compound's ability to undergo further chemical modifications makes it an essential tool for medicinal chemists working on the design and synthesis of new therapeutic agents. Beyond its use in synthesis, sodium nitromalonaldehyde is also important in the study of reactive intermediates. Due to its high reactivity, it is often used as a model compound in research focused on understanding the behavior of nitro-aldehyde systems. Researchers investigate its reactivity patterns to gain insights into reaction mechanisms and to develop new synthetic methodologies. This research is crucial for advancing the field of organic chemistry and for discovering new reactions that can be applied in various industries. In addition to its applications in synthesis and research, sodium nitromalonaldehyde has potential uses in materials science. Its reactive nature allows it to be incorporated into polymers, where it can influence the material's properties, such as stability and reactivity. This makes it a candidate for developing advanced materials with specialized functions, particularly in the fields of coatings and adhesives. In conclusion, sodium nitromalonaldehyde is a highly reactive and versatile compound with significant applications in organic synthesis, pharmaceutical development, and materials science. Its discovery and subsequent use in various chemical processes underscore its importance as a tool for advancing both scientific research and industrial applications. As chemists continue to explore its potential, sodium nitromalonaldehyde is likely to remain a valuable compound in the field of chemistry. References 2009. Synthesis of Five-Membered Nitroazoles. Nitroazoles: Synthesis, Structure and Applications. DOI: 10.1007/978-0-387-98070-6_1 |
| Market Analysis Reports |