Online Database of Chemicals from Around the World
| Changzhou Jiuwu Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiuwuchem.com | |||
 | +86 (519) 8512-1086 +86 13775162768 | |||
 | +86 (519) 8512-5369 | |||
 | info@jiuwuchem.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2016 | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic alcohol |
|---|---|
| Name | 4-(Trifluoromethyl)benzyl alcohol |
| Synonyms | [4-(trifluoromethyl)phenyl]methanol |
| Molecular Structure | 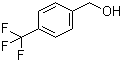 |
| Molecular Formula | C8H7F3O |
| Molecular Weight | 176.14 |
| CAS Registry Number | 349-95-1 |
| EC Number | 206-494-6 |
| SMILES | C1=CC(=CC=C1CO)C(F)(F)F |
| Density | 1.3±0.1 g/cm3, Calc.*, 1.286 g/mL (Expl.), 1.459 (Expl.) |
|---|---|
| Melting point | 18-20 °C (Expl.) |
| Index of Refraction | 1.463, Calc.* |
| Boiling Point | 213.7±0.0 °C (760 mmHg), Calc.*, 78-80 °C (4 mmHg) (Expl.) |
| Flash Point | 100.6±0.0 °C, Calc.*, 100 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
4-(Trifluoromethyl)benzyl alcohol is a chemical compound with the molecular formula C7H7F3O. It consists of a benzyl alcohol structure where a trifluoromethyl group (-CF3) is attached at the para position (position 4) on the aromatic ring. This compound belongs to the class of fluorinated aromatic alcohols, which are known for their unique chemical and physical properties, particularly their increased lipophilicity and potential for stability in harsh conditions. The discovery and synthesis of 4-(trifluoromethyl)benzyl alcohol are attributed to the interest in developing fluorinated compounds with enhanced reactivity and specific functionality in a wide range of chemical applications. The synthesis of 4-(trifluoromethyl)benzyl alcohol generally involves a two-step process starting with the formation of a trifluoromethylated benzyl intermediate, followed by selective reduction of the benzyl group to an alcohol. One of the common methods of introducing the trifluoromethyl group involves electrophilic aromatic substitution reactions, using reagents like trifluoromethyl iodide or a trifluoromethylating reagent under appropriate reaction conditions. The final alcohol is obtained through reduction of the corresponding benzyl aldehyde or ketone using reducing agents such as sodium borohydride or catalytic hydrogenation. 4-(Trifluoromethyl)benzyl alcohol has gained attention for its application in various fields, including organic synthesis, material science, and medicinal chemistry. One of the key reasons for its importance is the presence of the trifluoromethyl group, which significantly influences the compound's electronic properties, making it a valuable building block for the synthesis of more complex molecules. The trifluoromethyl group is electron-withdrawing, which affects the reactivity of the aromatic ring, often enhancing its ability to participate in nucleophilic substitution reactions and altering the reactivity of the alcohol group. In medicinal chemistry, 4-(trifluoromethyl)benzyl alcohol has been explored as a precursor for the development of pharmaceutical compounds. The trifluoromethyl group is known to increase the lipophilicity and metabolic stability of molecules, properties that are desirable in the development of drug candidates. Fluorinated aromatic alcohols, like 4-(trifluoromethyl)benzyl alcohol, are often used in the synthesis of biologically active compounds, including those with antimicrobial, anticancer, and anti-inflammatory activities. The presence of the alcohol group makes it a versatile intermediate for further functionalization, allowing for the attachment of a variety of bioactive groups. The compound is also used in the synthesis of advanced materials, such as fluorinated polymers and organic electronics. Fluorinated compounds like 4-(trifluoromethyl)benzyl alcohol have special properties, such as increased chemical resistance and improved thermal stability, making them suitable for use in applications where durability and resilience are important. For example, it is used in the preparation of fluorinated oligomers and monomers that are incorporated into high-performance materials like coatings, adhesives, and electronic devices. Furthermore, 4-(trifluoromethyl)benzyl alcohol is a useful intermediate in organic synthesis, especially in the preparation of complex fluorinated organic compounds. It serves as a key starting material for the synthesis of other fluorinated benzyl alcohol derivatives, which can be further modified to produce a wide range of chemical products, from industrial chemicals to specialty materials. The versatility of this compound as a reagent in synthetic chemistry highlights its importance in the broader field of fluorine chemistry. In conclusion, 4-(trifluoromethyl)benzyl alcohol is an important compound with diverse applications in chemical synthesis, medicinal chemistry, and material science. Its unique trifluoromethyl group imparts valuable electronic and physical properties that make it a key intermediate for the development of new pharmaceuticals and functional materials. Ongoing research into its reactivity and applications continues to unlock new possibilities for this versatile compound. References 2016. Synthesis of Trifluoromethylated Compounds. Journal of Fluorine Chemistry, 191. DOI: 10.1016/j.jfluchem.2016.09.008 |
| Market Analysis Reports |