Online Database of Chemicals from Around the World
| Hangzhou Qichuang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qc-chemical.com | |||
 | +86 (571) 8893-5129 | |||
 | +86 (571) 8825-0182 | |||
 | david@qc-chemical.com sales@qc-chemical.com davidw0828@gmail.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic alcohol |
|---|---|
| Name | 6,7-Dimethoxy-2-methylquinazolin-4-ol |
| Synonyms | 6,7-Dimethoxy-2-methylquinazolin-4(3H)-one; NSC 338200 |
| Molecular Structure | 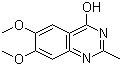 |
| Molecular Formula | C11H12N2O3 |
| Molecular Weight | 220.22 |
| CAS Registry Number | 35241-23-7 |
| SMILES | CC1=NC2=CC(=C(C=C2C(=O)N1)OC)OC |
| Density | 1.3±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 309.6±42.0 °C 760 mmHg (Calc.)* |
| Flash point | 141.1±27.9 °C (Calc.)* |
| Index of refraction | 1.616 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
6,7-Dimethoxy-2-methylquinazolin-4-ol is an organic compound belonging to the quinazoline class, which is a type of heterocyclic compound containing a fused benzene ring and a pyrimidine ring. This compound specifically features methoxy groups at positions 6 and 7, a methyl group at position 2, and a hydroxyl group at position 4 of the quinazoline ring. The methoxy groups (–OCH₃) are known to influence the compound’s solubility, electronic properties, and interactions with other molecules, making them significant in the context of drug design and material science. The quinazoline scaffold is an important structural motif in medicinal chemistry due to its broad range of biological activities. Quinazoline derivatives have been studied extensively for their potential therapeutic effects, particularly in the treatment of cancer, neurological disorders, and cardiovascular diseases. The presence of both electron-donating methoxy groups and a hydroxyl group in the structure of 6,7-dimethoxy-2-methylquinazolin-4-ol suggests that this compound could interact with a variety of biological targets, possibly through hydrogen bonding or electrostatic interactions, making it a valuable candidate for further study in drug development. The compound’s structure suggests that it may function as an inhibitor or modulator of specific enzymes or receptors, particularly those involved in signal transduction pathways that regulate cell growth and survival. Quinazoline derivatives are known to target protein kinases, which play a key role in cellular processes such as cell division, migration, and apoptosis. Additionally, the presence of the hydroxyl group may enable this compound to engage in hydrogen bonding interactions with biological targets, enhancing its ability to interact with biomolecules in a selective and potent manner. 6,7-Dimethoxy-2-methylquinazolin-4-ol could also be explored for its potential activity as an antimicrobial, antifungal, or anti-inflammatory agent. Quinazoline derivatives have shown promise in these areas due to their ability to interfere with cellular processes critical for the survival of pathogens. The methoxy and hydroxyl substitutions on the aromatic ring may increase the compound’s ability to cross biological membranes and interact with intracellular targets, further enhancing its potential as a therapeutic agent. In addition to its biological applications, the compound could be studied for its potential use in materials science. Quinazoline derivatives are sometimes used as components in organic electronics, such as organic light-emitting diodes (OLEDs) and organic semiconductors, due to their ability to exhibit interesting electronic properties. The methoxy groups in particular can modify the electronic characteristics of the quinazoline scaffold, allowing for fine-tuning of its behavior in electronic devices. The presence of the methyl group at position 2 of the quinazoline ring may further influence the compound’s electronic properties, steric effects, and its interaction with receptors or enzymes. The introduction of such methylation could enhance lipophilicity, potentially improving the compound’s ability to enter cells or cross the blood-brain barrier, which is particularly useful in the development of drugs for neurological diseases. 6,7-Dimethoxy-2-methylquinazolin-4-ol is also of interest in the context of synthetic chemistry, where it may serve as a valuable intermediate in the preparation of more complex compounds. Quinazoline derivatives can be modified by introducing additional functional groups at various positions on the aromatic and heterocyclic rings, thereby expanding the diversity of molecules that can be synthesized for research and drug development. In summary, 6,7-dimethoxy-2-methylquinazolin-4-ol is a versatile compound with potential applications in medicinal chemistry, materials science, and synthetic chemistry. Its structural features, including methoxy and hydroxyl groups, make it a promising candidate for the development of bioactive molecules with a wide range of therapeutic activities. Whether used as a kinase inhibitor, antimicrobial agent, or as part of organic electronic devices, this quinazoline derivative offers significant potential for further investigation and development in various scientific fields. |
| Market Analysis Reports |