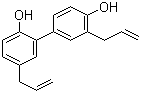
Honokiol is a naturally occurring biphenolic compound isolated primarily from the bark, seed cones, and leaves of trees in the genus Magnolia. It is a low-molecular-weight lignan-type polyphenol with the molecular formula C18H18O2. Structurally, it consists of two phenyl rings linked through a carbon–carbon bond, with each aromatic ring bearing hydroxyl and allyl substituents.
The core structure of honokiol is a biphenyl system, which provides a rigid but rotatable framework between the two aromatic rings. Each ring contains a hydroxyl group (–OH) and an allyl substituent (–CH2CH=CH2) positioned para or ortho depending on the substitution pattern. This combination of phenolic and unsaturated side-chain functionality contributes to its chemical reactivity and biological interactions.
Honokiol is biosynthesized in Magnolia species through the phenylpropanoid pathway, a major plant metabolic route responsible for the formation of many phenolic natural products. In this pathway, cinnamic acid derivatives are enzymatically transformed into phenolic intermediates, which are then coupled and modified to form biphenyl lignans such as honokiol and its structural isomer magnolol.
The compound was first isolated and characterized from Magnolia bark extracts, which have been used in traditional East Asian herbal medicine for centuries. Although traditional use predates its chemical identification, modern phytochemical studies have established honokiol as one of the principal bioactive constituents of Magnolia officinalis extracts.
Chemically, the phenolic hydroxyl groups in honokiol are capable of hydrogen bonding and can undergo typical phenol reactions such as esterification, etherification, and oxidation. The allyl side chains contain carbon–carbon double bonds that can participate in electrophilic addition reactions, oxidation, or polymerization under suitable conditions. The biphenyl structure also allows for conformational flexibility due to rotation around the central C–C bond.
Honokiol is relatively lipophilic compared with many other polyphenols, due to the presence of nonpolar allyl groups and the absence of extensive sugar or polar substituents. However, the phenolic hydroxyl groups impart some polarity and enable interaction with aqueous environments and biological macromolecules.
In pharmacological research, honokiol has been extensively studied for a variety of biological activities. Investigations have examined its interactions with cellular signaling pathways, including modulation of inflammatory mediators, oxidative stress responses, and apoptosis-related mechanisms. It has also been studied in the context of neurological, cardiovascular, and anticancer research models.
The compound can interact with proteins, enzymes, and lipid membranes due to its amphiphilic character. The phenolic groups can engage in hydrogen bonding and radical scavenging, while the hydrophobic biphenyl core facilitates membrane association. These properties contribute to its broad range of observed biological effects in experimental systems.
From a chemical synthesis perspective, honokiol can be obtained through isolation from plant sources or prepared via synthetic routes involving biphenyl coupling and selective functionalization of phenolic precursors. Laboratory synthesis often focuses on constructing the biphenyl backbone followed by installation of hydroxyl and allyl substituents.
Physicochemically, honokiol is a crystalline solid with limited water solubility but good solubility in organic solvents. Its conjugated aromatic system gives rise to characteristic ultraviolet absorption properties typical of phenolic compounds.
Historically, honokiol has been of interest both as a traditional medicinal constituent and as a modern pharmacological research compound. Its identification as a major active component of Magnolia extracts has driven extensive chemical and biological investigation.
Overall, honokiol is a naturally derived biphenolic compound with a substituted biphenyl core containing hydroxyl and allyl groups. Its chemical structure underlies a range of interactions with biological systems and has made it an important subject in natural products chemistry and biomedical research.
References
2026. Nano Based Drug Delivery Formulations for the Prevention and Treatment of Dental Caries. Current Oral Health Reports.
DOI: 10.1007/s40496-026-00423-z
2026. Honokiol attenuates renal fibrosis via SIRT3-Mediated regulation of Wnt/β-Catenin signaling. Functional & Integrative Genomics.
DOI: 10.1007/s10142-025-01816-6
2026. Honokiol blocks tumor development and metastasis through mitochondrion-targeted effects. Cell Death & Disease.
URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12877151
|





















































































 GHS05;GHS07;GHS09 Danger Details
GHS05;GHS07;GHS09 Danger Details