Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Beijing Huikang Boyuan Chemical Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huikangchem.com | |||
 | +86 (10) 6886-2197 6886-7502 | |||
 | +86 (10) 6888-2204 | |||
 | market@huikangchem.com sales@huikangchem.com sales3@huikangchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Beijing Reco Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.recotechnology.com | |||
 | +86 (10) 6060-5164 | |||
 | +86 (10) 6060-5164 | |||
 | sales@recotechnology.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Nanjing Bic Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bicbiotech.com | |||
 | +86 18851183712 | |||
 | sales@bicbiotech.com | |||
 | QQ Chat | |||
 | WeChat: +8618851183712 | |||
 | WhatsApp:+8618851183712 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Imidazoles |
|---|---|
| Name | 1-Methyl-5-nitro-1H-benzimidazole-2-butanoic acid ethyl ester |
| Synonyms | ethyl 4-(1-methyl-5-nitrobenzimidazol-2-yl)butanoate |
| Molecular Structure | 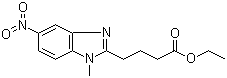 |
| Molecular Formula | C14H17N3O4 |
| Molecular Weight | 291.30 |
| CAS Registry Number | 3543-72-4 |
| EC Number | 686-742-1 |
| SMILES | CCOC(=O)CCCC1=NC2=C(N1C)C=CC(=C2)[N+](=O)[O-] |
| Density | 1.4$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 577.4$+/-$50.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 303.0$+/-$30.1 $degree$C (Calc.)* |
| Index of refraction | 1.734 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS06;GHS07;GHS08 Danger Details GHS06;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H302-H312-H315-H319-H332-H341-H351 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P261-P264-P264+P265-P270-P271-P280-P301+P316-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P318-P321-P330-P332+P317-P337+P317-P362+P364-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
1-Methyl-5-nitro-1H-benzimidazole-2-butanoic acid ethyl ester is a functionalized benzimidazole derivative featuring a nitro-substituted aromatic ring, a butanoic acid side chain, and an ethyl ester functionality. Benzimidazole compounds have been widely studied since the early 20th century for their biological activity, chemical stability, and structural versatility. The presence of the 5-nitro group and the 1-methyl substitution enhances electronic and steric properties, while the ethyl ester of the butanoic acid provides a reactive site for hydrolysis or further derivatization, making this molecule valuable in medicinal chemistry and synthetic applications. Structurally, the molecule consists of a fused bicyclic benzimidazole ring with a methyl group attached to the nitrogen at position 1 and a nitro group at the 5-position. At position 2, a butanoic acid chain is esterified with ethanol, forming an ethyl ester. The nitro group is an electron-withdrawing substituent that affects the electronic density of the aromatic ring, which can influence reactivity in electrophilic and nucleophilic aromatic substitution reactions. The ethyl ester allows controlled hydrolysis to the free carboxylic acid or conversion to amides and other derivatives, providing versatility for synthetic modification or drug design. The synthesis of 1-methyl-5-nitro-1H-benzimidazole-2-butanoic acid ethyl ester typically starts from 5-nitro-1-methylbenzimidazole or its derivatives. The 2-position is functionalized with a butanoic acid chain via alkylation or acylation reactions. Subsequent esterification with ethanol under acidic or catalytic conditions produces the ethyl ester. These synthetic strategies allow selective functionalization at the nitrogen, nitro-substituted aromatic ring, and carboxylic acid, providing a high-purity intermediate suitable for further chemical or pharmaceutical applications. In medicinal chemistry, nitro-substituted benzimidazole derivatives such as this compound are of interest due to their potential antimicrobial, antiviral, or enzyme inhibitory activities. The benzimidazole ring mimics purine bases, allowing interactions with nucleic acid-processing enzymes, and the nitro group can participate in redox reactions, which are relevant in antimicrobial mechanisms. The ethyl ester moiety may be used as a prodrug strategy, facilitating cellular uptake and controlled release of the active acid form in vivo. Beyond pharmacological applications, this compound serves as a versatile intermediate in organic synthesis. The ethyl ester group can be hydrolyzed to yield the carboxylic acid for peptide coupling, amide formation, or other conjugation reactions. The nitro group is chemically reactive, enabling reduction to an amino group or participation in substitution reactions to introduce additional functionality. The fused benzimidazole scaffold provides rigidity, aromaticity, and planarity, which are useful features in the design of heterocyclic libraries and structural analogs. Overall, 1-methyl-5-nitro-1H-benzimidazole-2-butanoic acid ethyl ester represents a multifunctional benzimidazole derivative combining a nitro-substituted aromatic system, a methylated nitrogen, and a carboxylic ester side chain. Its chemical features offer versatility for synthesis, derivatization, and exploration of biological activity, making it a valuable compound in medicinal chemistry, heterocyclic chemistry, and the development of bioactive small molecules. References 2007. Bendamustine hydrochloride. Pharmaceutical Substances. PubChem Literature ID: 906195298 |
| Market Analysis Reports |