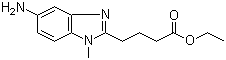
1-Methyl-5-amino-1H-benzimidazole-2-butanoic acid ethyl ester is a functionalized benzimidazole derivative in which the 5-position of the aromatic ring carries an amino group, the 1-position nitrogen is methylated, and the 2-position bears a butanoic acid side chain esterified as an ethyl ester. Benzimidazole scaffolds are widely recognized in medicinal chemistry for their stability, planarity, and ability to participate in hydrogen bonding, π–π interactions, and coordination with biological macromolecules. The presence of the amino group at the 5-position increases hydrogen-bonding potential and nucleophilicity, enhancing the molecule’s versatility for chemical modification and biological interactions.
Structurally, the compound consists of a fused bicyclic benzimidazole ring system, with a methyl group on the N1 nitrogen to modify steric and electronic properties, and a 5-amino substituent on the aromatic ring. The ethyl ester of the 2-butanoic acid side chain provides a functional handle for hydrolysis to the free carboxylic acid or for formation of amides and other derivatives. The combination of the amino group, ester, and methylation allows chemists to selectively target different positions for synthetic modifications, while the planar bicyclic core ensures favorable stacking and binding interactions in biological systems.
The synthesis of 1-methyl-5-amino-1H-benzimidazole-2-butanoic acid ethyl ester generally starts with 5-nitro-1-methylbenzimidazole derivatives. The nitro group is reduced under controlled conditions to yield the amino functionality. Introduction of the butanoic acid side chain at the 2-position can be achieved via alkylation or acylation reactions, followed by esterification with ethanol to form the ethyl ester. These synthetic steps allow selective functionalization while preserving the benzimidazole core, producing high-purity intermediates suitable for medicinal or synthetic applications.
In medicinal chemistry, 5-amino-substituted benzimidazoles are explored as potential enzyme inhibitors, receptor modulators, and antimicrobial agents. The amino group can engage in hydrogen bonding with enzyme active sites or receptor residues, improving binding affinity and selectivity. The methyl group at the nitrogen may enhance metabolic stability and influence lipophilicity, while the ester side chain can act as a prodrug motif, allowing controlled in vivo hydrolysis to release the active carboxylic acid form. This combination of structural features makes the molecule suitable for the development of bioactive small molecules and peptide conjugates.
Beyond pharmacological applications, 1-methyl-5-amino-1H-benzimidazole-2-butanoic acid ethyl ester serves as a versatile intermediate in synthetic chemistry. The amino group can be derivatized through acylation, sulfonylation, or alkylation, while the ester group can be transformed into amides, acids, or other conjugates. The benzimidazole core is stable under various reaction conditions and can participate in electrophilic or nucleophilic substitution reactions, allowing the construction of more complex heterocyclic systems.
Overall, 1-methyl-5-amino-1H-benzimidazole-2-butanoic acid ethyl ester exemplifies a multifunctional benzimidazole derivative with a reactive amino group, a methylated nitrogen, and an esterified side chain. Its combination of chemical versatility, hydrogen-bonding capacity, and structural rigidity makes it valuable for synthetic transformations, medicinal chemistry, and the development of biologically active compounds.
References
2007. Bendamustine hydrochloride. Pharmaceutical Substances.
PubChem Literature ID: 906195298
|






























































 GHS06;GHS07;GHS08 Danger Details
GHS06;GHS07;GHS08 Danger Details