2,6-Dichlorobenzoxazole is a chemical compound belonging to the class of benzoxazoles, characterized by a fused benzene and oxazole ring system. This compound has attracted attention in both research and industrial applications due to its unique structure and properties. The discovery of 2,6-dichlorobenzoxazole can be traced back to the early 20th century when chemists were exploring various heterocyclic compounds. The introduction of chlorine atoms at the 2 and 6 positions of the benzoxazole ring enhances its reactivity and opens up avenues for further chemical modifications.
The synthesis of 2,6-dichlorobenzoxazole typically involves the chlorination of benzoxazole or its derivatives, which can be achieved through various methods, including electrophilic aromatic substitution reactions. The presence of chlorine atoms in the molecule not only influences its chemical reactivity but also contributes to its solubility and stability, making it a valuable intermediate in organic synthesis.
One of the primary applications of 2,6-dichlorobenzoxazole is in the pharmaceutical industry, where it serves as a key building block for the synthesis of various bioactive compounds. Its derivatives have been explored for their potential therapeutic properties, including antibacterial and antifungal activities. Research has indicated that modifications to the benzoxazole structure can lead to compounds with enhanced efficacy against a range of pathogens. This makes 2,6-dichlorobenzoxazole a valuable precursor in the development of new antimicrobial agents.
In addition to its pharmaceutical applications, 2,6-dichlorobenzoxazole has found utility in materials science. The compound can be incorporated into polymeric materials to impart specific properties, such as increased thermal stability and improved mechanical performance. Its unique chemical structure allows for the development of advanced materials with tailored characteristics, making it relevant in the production of coatings, adhesives, and other specialty products.
Furthermore, 2,6-dichlorobenzoxazole has potential applications in agrochemicals, particularly as a component in the synthesis of herbicides and fungicides. The compound's reactivity and ability to interact with biological systems position it as a candidate for developing effective agrochemical formulations. Ongoing research aims to explore these applications further, focusing on optimizing the efficacy and environmental safety of related compounds.
In summary, 2,6-dichlorobenzoxazole is a versatile chemical compound with significant implications in pharmaceutical development, materials science, and agrochemicals. Its discovery and subsequent applications highlight the importance of heterocyclic compounds in various fields of research and industry. As scientists continue to explore the potential of 2,6-dichlorobenzoxazole and its derivatives, the compound is expected to play a crucial role in the advancement of new therapeutic agents and innovative materials.
References
2020. Experimental Measurement and Correlation of Solubilities of R-2[4-(6-Chloro-2-Benzoxazolyloxy)phenoxy]propanoic Acid in Methanol, Ethanol, and Methanol-Ethanol Mixed Solvents. Russian Journal of Physical Chemistry A, 94(13).
DOI: 10.1134/s0036024420130312
2016. Synthesis and herbicidal activity of novel N-allyloxy/propargyloxy aryloxyphenoxy propionamide derivatives. Chemical Research in Chinese Universities, 32(3).
DOI: 10.1007/s40242-016-5415-0
2022. Cross-Coupling Reactions of Polyhalogenated Heterocycles. Synlett, 33(10).
DOI: 10.1055/s-0040-1719906
|












































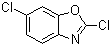



 GHS05;GHS06;GHS07;GHS09 Danger Details
GHS05;GHS06;GHS07;GHS09 Danger Details