Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Biochemical >> Biochemical reagent >> Acid-base indicator |
|---|---|
| Name | 2,2'-Dipyridyl |
| Synonyms | 2,2'-Bipyridine |
| Molecular Structure | 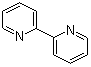 |
| Molecular Formula | C10H8N2 |
| Molecular Weight | 156.19 |
| CAS Registry Number | 366-18-7 |
| EC Number | 206-674-4 |
| SMILES | C1=CC=NC(=C1)C2=CC=CC=N2 |
| Water solubility | 5.5 g/L (22 °C) |
|---|---|
| Density | 1.1±0.1 g/cm3, Calc.* |
| Melting point | 70-73 °C (Expl.) |
| Index of Refraction | 1.581, Calc.* |
| Boiling Point | 272.5±0.0 °C (760 mmHg), Calc.*, 273 °C (Expl.) |
| Flash Point | 107.2±12.0 °C, Calc.*, 121 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS06;GHS07 Danger Details GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301+H311-H301-H311-H312-H319-H412 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P262-P264-P264+P265-P270-P273-P280-P301+P316-P302+P352-P305+P351+P338-P316-P317-P321-P330-P337+P317-P361+P364-P362+P364-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1663; UN 2811 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
2,2'-Dipyridyl, also known as bipyridine, is a chemical compound with the formula C10H8N2. It consists of two pyridine rings (a six-membered ring containing nitrogen atoms) that are connected by a single bond at the 2-position. This compound is a versatile ligand in coordination chemistry and has various applications in materials science, catalysis, and biological studies. The discovery of 2,2'-dipyridyl dates back to the early 19th century, with initial work focusing on its synthesis and its properties as a simple organic molecule. Since then, it has gained prominence due to its ability to coordinate with metal ions, forming stable complexes. These metal complexes are of great interest in the field of organometallic chemistry and catalysis. The ability of 2,2'-dipyridyl to donate electron density through the nitrogen atoms allows it to act as a bidentate ligand, which makes it particularly useful in the synthesis of various metal-organic frameworks (MOFs) and in the design of catalysts. One of the major applications of 2,2'-dipyridyl is in the field of coordination chemistry, where it is used to form coordination complexes with a wide range of transition metals. These metal complexes have been studied for their role in catalysis, particularly in reactions such as hydrogenation, oxidation, and polymerization. The flexibility and stability of 2,2'-dipyridyl make it an ideal ligand for the design of new catalysts that can facilitate various chemical transformations. In addition to its use in catalysis, 2,2'-dipyridyl is also widely employed in the field of material science, especially in the preparation of metal-organic frameworks (MOFs). MOFs are porous materials with a wide range of applications, including gas storage, sensing, and drug delivery. The ability of 2,2'-dipyridyl to bind with metal ions and form stable structures is crucial for the development of these materials, as it provides the necessary framework for the incorporation of different metal ions and guest molecules. 2,2'-Dipyridyl has also found applications in biological systems, where it is used as a chelating agent for metal ions in various biochemical and pharmacological studies. It has been used to study metal ion interactions with proteins, enzymes, and other biomolecules. Furthermore, 2,2'-dipyridyl and its metal complexes have been investigated for their potential use in medicinal chemistry, particularly in the development of metal-based drugs for treating diseases like cancer and Alzheimer’s disease. References 2018. Bipyridine Ligands in Coordination Chemistry. Coordination Chemistry Reviews, 374. DOI: 10.1016/j.ccr.2018.06.003 |
| Market Analysis Reports |