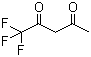
1,1,1-Trifluoro-2,4-pentanedione (C5HF3O2) is a fluorinated β-diketone compound that contains both a trifluoromethyl group (–CF3) and two carbonyl groups. This compound belongs to the class of fluorinated organic compounds, which are recognized for their stability and unique reactivity, owing to the presence of fluorine atoms. It has been widely studied for its potential applications in various fields, including organic synthesis, materials science, and as a precursor for other fluorinated compounds.
The discovery of 1,1,1-trifluoro-2,4-pentanedione stems from the growing interest in fluorinated β-diketones, as these compounds are valuable intermediates in organic chemistry due to their reactivity. The trifluoromethyl group significantly enhances the electrophilicity of the carbonyl carbon atoms, which makes the compound an excellent candidate for various chemical reactions, such as nucleophilic addition or condensation reactions. The structure of 1,1,1-trifluoro-2,4-pentanedione consists of a pentanedione backbone with a trifluoromethyl group attached to the first carbon, which makes it a highly functional compound for synthetic purposes.
1,1,1-Trifluoro-2,4-pentanedione has found several important applications, primarily in the synthesis of fluorinated metal complexes, as a reagent in organic synthesis, and in the preparation of specialty chemicals. One notable application is its use in the preparation of metal trifluoroacetate complexes. The β-diketone group in 1,1,1-trifluoro-2,4-pentanedione can form chelates with various metal ions, such as those of copper, nickel, and lanthanides. These metal complexes have been studied for their potential in catalysis and in materials science, where they can be used as catalysts in various reactions, including polymerization and oxidation processes.
In addition to its use in metal complexation, 1,1,1-trifluoro-2,4-pentanedione serves as a valuable reagent in the synthesis of fluorinated organic molecules. The trifluoromethyl group enhances the reactivity of the diketone, making it suitable for reactions like the Michael addition, where it can react with nucleophiles to form fluorinated carbon-carbon bonds. This ability to form fluorinated intermediates is essential in the production of fluorinated pharmaceuticals, agrochemicals, and other specialty chemicals.
Another significant application of 1,1,1-trifluoro-2,4-pentanedione is its role as a precursor in the production of fluorinated polymers. The compound can be polymerized or used in the synthesis of monomers that contain the trifluoromethyl group, which imparts unique properties to the resulting polymer, such as high chemical resistance, stability at high temperatures, and low surface energy. Fluorinated polymers are highly sought after in industries such as electronics, automotive, and aerospace, where materials with superior thermal and chemical stability are required.
Additionally, 1,1,1-trifluoro-2,4-pentanedione has been investigated for its potential use in the field of organic electronics, particularly in the fabrication of organic semiconductors. The trifluoromethyl group in the compound can enhance the electronic properties of organic materials, which may improve the performance of organic light-emitting diodes (OLEDs), organic solar cells, and field-effect transistors (OFETs).
The compound’s chemical stability, resistance to heat, and low volatility also make it useful in the production of coatings and surface treatments, where it can provide improved resistance to harsh environmental conditions.
Given the potential environmental and health concerns associated with fluorinated compounds, the use of 1,1,1-trifluoro-2,4-pentanedione must be carefully managed. Fluorinated compounds tend to persist in the environment, and their potential accumulation could pose risks to ecosystems. Thus, regulations regarding the safe handling and disposal of these chemicals are crucial to minimize their environmental impact.
In conclusion, 1,1,1-trifluoro-2,4-pentanedione is a versatile and important fluorinated β-diketone compound. It has applications in metal complexation, organic synthesis, polymer production, and organic electronics. The trifluoromethyl group in the compound imparts unique chemical reactivity, making it a valuable reagent for the development of fluorinated materials and specialty chemicals. However, its environmental impact must be carefully monitored to ensure sustainable use in industrial and chemical processes.
References
2024. Synthesis and Structure of Ag(I) Complexes with Tris(3,5-Dimethylpyrazol-1-yl)Methane and β-Diketones. Journal of Structural Chemistry, 65(11).
DOI: 10.1134/s0022476624110180
2023. Revisiting photoisomerization in fluorinated analogues of acetylacetone trapped in cryogenic matrices. The European Physical Journal D, 77(8).
DOI: 10.1140/epjd/s10053-023-00727-0
2022. Synthesis, Characterization and Single Crystal X-Ray Structures of Trifluroacetylacetonate Copper(II) Complexes. Journal of Chemical Crystallography, 52(4).
DOI: 10.1007/s10870-022-00937-1
|






























































 GHS02;GHS07 Warning Details
GHS02;GHS07 Warning Details