Online Database of Chemicals from Around the World
| Lanzhou Kohn & Shawn Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kyxpharma.com | |||
 | +86 (931) 419-0454 +86 13893344397 | |||
 | +86 (931) 850-6154 | |||
 | sales@kyxpharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrazines |
|---|---|
| Name | 2-Chloro-3,5-dimethylpyrazine |
| Molecular Structure | 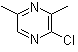 |
| Molecular Formula | C6H7ClN2 |
| Molecular Weight | 142.59 |
| CAS Registry Number | 38557-72-1 |
| EC Number | 696-037-0 |
| SMILES | CC1=CN=C(C(=N1)C)Cl |
| Solubility | Sparingly soluble (21 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.184±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Boiling point | 186.6±35.0 °C (760 Torr), Calc.* |
| Flash point | 83.1±11.5 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2013 ACD/Labs) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P312-P330-P332+P313-P337+P313-P362-P363-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
2-Chloro-3,5-dimethylpyrazine is a compound of great interest due to its wide range of applications in industries ranging from pharmaceuticals to food and flavors. 2-Chloro-3,5-dimethylpyrazine was synthesized and characterized for the first time by the reaction of 2,3-dimethylpyrazine with chlorine gas or a chlorinating agent. This synthesis marked a major advance in heterocyclic chemistry and provided a route to chlorinated pyrazine derivatives with unique chemical properties. The molecular structure of 2-chloro-3,5-dimethylpyrazine consists of a pyrazine ring with a chlorine substituent at the 2-position and methyl groups at the 3- and 5-positions. This arrangement gives it specific chemical properties, including moderate volatility and a unique aroma. The chlorine groups enhance its reactivity, making it valuable in a variety of chemical transformations and applications. In the flavor and fragrance industry, 2-chloro-3,5-dimethylpyrazine is highly valued for its nutty, roasted, and earthy aromas. It is commonly used as a flavoring agent in foods such as baked goods, savory snacks, and beverages. Its characteristic odor helps enhance the sensory appeal of these products, providing a rich and complex flavor profile. In the pharmaceutical field, 2-chloro-3,5-dimethylpyrazine is a key intermediate in the synthesis of drug compounds. Its functional groups are able to modify the bioactivity and pharmacokinetic properties of drug molecules. Researchers have explored the potential application of its derivatives in drug discovery programs, focusing on its role in the development of therapeutic agents for various medical conditions. From a chemical perspective, 2-chloro-3,5-dimethylpyrazine can be used as a building block in organic synthesis. Its reactivity allows the introduction of various functional groups, which helps create novel compounds with customized properties. It is a versatile precursor for the synthesis of complex molecules, including agrochemicals, dyes, and materials used in electronic applications. Research on 2-chloro-3,5-dimethylpyrazine continues to grow, thanks to its unique chemical properties and wide range of application potential. Future research aims to explore its environmental impact, develop more efficient synthesis methods, and expand its application in emerging fields such as nanotechnology and biotechnology. References 2018. Alkyl-Dimethylpyrazines in Mandibular Gland Secretions of Four Odontomachus Ant Species (Formicidae: Ponerinae). Journal of Chemical Ecology, 44(3). DOI: 10.1007/s10886-018-0948-y 2014. Characterization and analysis of structural isomers of dimethyl methoxypyrazines in cork stoppers and ladybugs (Harmonia axyridis and Coccinella septempunctata). Analytical and Bioanalytical Chemistry, 406(22). DOI: 10.1007/s00216-014-8049-4 2004. Deoxidative Chlorination of Pyrazine N-Oxides. Science of Synthesis. URL: SD-016-00973 |
| Market Analysis Reports |