Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Futoh Chemicals Co., Ltd. | Japan | |||
|---|---|---|---|---|
 | www.futoh.co.jp | |||
 | +81 (52) 521-8171 | |||
 | +81 (52) 521-8793 | |||
 | tokoro@futoh.co.jp | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Ring Specialty Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.ringchemicals.com | |||
 | +1 (416) 493-6870 | |||
 | info@ringchemicals.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Liaoning Hongtai Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lnhtbio.com | |||
 | +86 (24) 7882-6666 | |||
 | +86 (24) 7882-6999 | |||
 | hongtai.bio@vip.163.com | |||
| Chemical manufacturer since 2000 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Kingreat Chemistry Company Limited | China | |||
|---|---|---|---|---|
 | www.chemtrade.cn | |||
 | +86 (592) 229-9609 | |||
 | +86 (592) 229-9601 | |||
 | kingreat@vip.163.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Kessie Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kessiechem.com | |||
 | +86 (519) 8698-7916 +86 15851999766 | |||
 | +86 (519) 8690-8212 | |||
 | info@kessiechem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Finetech Industry Limited | China | |||
|---|---|---|---|---|
 | www.finetechchem.com | |||
 | +86 (27) 8746-5837 +86 18971612321 | |||
 | +86 (27) 8777-2287 | |||
 | sales@finetechnology-ind.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| EnBridge PharmTech (Wuxi) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enbridgepharm.com | |||
 | +86 (510) 8359-1909 +86 18018392058 | |||
 | +86 (510) 8359-1909 | |||
 | info@enbridgepharm.com | |||
 | QQ Chat | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Cradlechem (Jiangsu) Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cradlechem.com | |||
 | +86 (519) 8571-5016 8503-3388 8571-6188 | |||
 | +86 (519) 8571-6288 | |||
 | cradlechem@163.com | |||
| Chemical manufacturer since 1988 | ||||
| Jintan Huayang Chemical Plant | China | |||
|---|---|---|---|---|
 | www.huayangchem.com | |||
 | +86 (519) 8252-1156 8252-7838 | |||
 | +86 (519) 8252-1118 | |||
 | hychem@huayangchem.com | |||
| Chemical manufacturer since 1992 | ||||
| Acros Organics | Belgium | |||
|---|---|---|---|---|
 | www.acros.com | |||
 | +86 (21) 5258-1100 | |||
 | +86 (21) 5258-0119 | |||
 | info@acros.com | |||
| Chemical manufacturer | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Trigona oHG | Germany | |||
|---|---|---|---|---|
 | www.trigona.de | |||
 | +49 (611) 962-5283 | |||
 | +49 (611) 962-9032 | |||
 | info@trigona.de | |||
| Chemical manufacturer | ||||
| Cambrex Corporation | USA | |||
|---|---|---|---|---|
 | www.cambrex.com | |||
 | +1 (201) 804-3000 | |||
 | +1 (201) 804-9852 | |||
 | info@cambrex.com | |||
| Chemical manufacturer | ||||
| Reuter Chemische Apparatebau KG | Germany | |||
|---|---|---|---|---|
 | www.rca-separations.de | |||
 | +49 (761) 559-6460 | |||
 | +49 (761) 559-6488 | |||
 | sales@rca-separations.de | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Imide |
|---|---|
| Name | (R)-(+)-1-Phenylethylamine |
| Synonyms | D-alpha-Phenylethylamine; R-(+)-alpha-Methylbenzylamine; D(+)-alpha-Methylbenzylamine; (+)-PEA; (R)-(+)-1-Phenylethylamine |
| Molecular Structure | 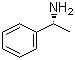 |
| Molecular Formula | C8H11N |
| Molecular Weight | 121.18 |
| CAS Registry Number | 3886-69-9 |
| EC Number | 223-423-4 |
| SMILES | C[C@H](C1=CC=CC=C1)N |
| Density | 1.0±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 32 °C (Expl.), 0.951 g/mL (Expl.) |
| Index of Refraction | 1.533, Calc.*,1.526 (Expl.) |
| Boiling Point | 183.0±9.0 °C (760 mmHg), Calc.*, 185.2 °C (Expl.) |
| Flash Point | 79.4 °C, Calc.*, 79 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H314-H318 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P260-P264-P264+P265-P270-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P330-P362+P364-P363-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| Transport Information | UN 2735 | ||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
(R)-(+)-1-Phenylethylamine is an organic compound that belongs to the class of amines and is a chiral molecule with important biological and chemical properties. It is the enantiomer of 1-phenylethylamine, which is a naturally occurring compound found in various plants and animals. The (R)-(+)-enantiomer, specifically, plays a significant role in several biological processes and has applications in various chemical and pharmaceutical fields. The compound was first synthesized in the early 20th century, with its structure and properties becoming more clearly understood through chemical analysis and synthetic advancements. It is typically obtained through enantioselective synthesis methods that yield the (R)-enantiomer, which has specific stereochemical properties distinguishing it from the (S)-enantiomer. The (R)-(+)-configuration is associated with particular pharmacological effects, which differ from those of the (S)-enantiomer and the racemic mixture of both enantiomers. (R)-(+)-1-Phenylethylamine is widely recognized for its role in the central nervous system. It acts as a neuromodulator and is chemically related to other important biogenic amines such as dopamine, norepinephrine, and serotonin. In the human body, (R)-(+)-1-phenylethylamine functions as a trace amine and is thought to influence mood, behavior, and cognitive function. It has been studied for its potential effects on mood regulation, alertness, and attention. The compound has garnered attention in the pharmaceutical industry due to its potential applications in drug development. It has been investigated for use in the treatment of mood disorders, such as depression and anxiety, owing to its ability to modulate neurotransmitter systems in the brain. Research suggests that (R)-(+)-1-phenylethylamine can influence the release and reuptake of monoamine neurotransmitters, particularly in the context of the central nervous system. Some studies have also explored its potential as a therapeutic agent for conditions such as attention deficit hyperactivity disorder (ADHD) and cognitive enhancement. In addition to its pharmacological uses, (R)-(+)-1-phenylethylamine is employed in the synthesis of various compounds, including pharmaceuticals, agrochemicals, and flavoring agents. It is used as a building block in the synthesis of other amines and derivatives that possess specific biological activities. Its ability to modify the structure and properties of other molecules makes it an important intermediate in the development of new chemical entities. In the realm of materials science, (R)-(+)-1-phenylethylamine has also been studied for its potential role in the preparation of chiral materials, particularly in asymmetric synthesis and catalysis. The presence of the chiral center in this molecule allows it to influence the stereochemistry of reactions, making it useful in the creation of molecules with specific three-dimensional arrangements, which are important in drug discovery and molecular design. The compound has also been investigated for its potential use in the food and fragrance industries. Due to its distinct aromatic properties, (R)-(+)-1-phenylethylamine is sometimes utilized as a flavoring agent in certain food products and in the creation of perfumes. It contributes to the fragrance profile of certain substances, where its aromatic and structural properties make it an attractive component in complex formulations. In conclusion, (R)-(+)-1-phenylethylamine is a versatile and important chemical compound with diverse applications in pharmaceutical development, materials science, and the fragrance industry. Its biological activities, including its potential to influence mood and cognitive function, make it a subject of ongoing research, particularly in the context of neurological and psychiatric disorders. Additionally, its role as a chiral building block in synthetic chemistry and its potential for use in various industrial applications highlight its significance in both academic and commercial fields. References 1996. Preparation of optically pure chiral amines by lipase-catalyzed enantioselective hydrolysis of N-acyl-amines. Biotechnology Techniques, 10(5). DOI: 10.1007/bf00173249 2022. Synthesis of Antimalarial BRD5018. Synfacts, 18(07). DOI: 10.1055/s-0041-1738389 2023. Optimization of the regioselective synthesis of mixed cellulose 3,5-dimethylphenyl and α-phenylethyl carbamate selectors as separation phases for chiral HPLC. Cellulose, 30(3). DOI: 10.1007/s10570-022-05007-5 |
| Market Analysis Reports |