Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| TH Chemica Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.thchemica.com | |||
 | +86 (573) 8283-5115 +86 13998887555 | |||
 | +86 (573) 8396-5652 | |||
 | thchemica@126.com info@thchemica.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Win-Win Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.win-winchemical.com | |||
 | +86 (577) 6449-8589 +86 15325081899 | |||
 | +86 (577) 5699-4596 | |||
 | sales@win-winchemical.com winwinchemical@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Aromsyn Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aromsyn.com | |||
 | +86 (571) 8558-5865 +86 13336024896 | |||
 | +86 (571) 8558-5865 | |||
 | service@aromsyn.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Marshallton Research Laboratories | USA | |||
|---|---|---|---|---|
 | www.marshalltonlabs.com | |||
 | +1 (336) 983-2131 | |||
 | +1 (336) 983-0096 | |||
 | marshallton@windstream.net | |||
| Chemical manufacturer since 1974 | ||||
| Melford Laboratories Limited | UK | |||
|---|---|---|---|---|
 | www.melford.co.uk | |||
 | +44 (1449) 741-178 | |||
 | +44 (1449) 741-217 | |||
 | pfranklin@melford.co.uk sales@melford.co.uk | |||
| Chemical manufacturer since 1985 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Ricci Chimica | Italy | |||
|---|---|---|---|---|
 | www.riccichimica.com | |||
 | +39 (75) 692-9241 | |||
 | +39 (75) 592-6595 | |||
 | info@riccichimica.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic acid |
|---|---|
| Name | 4-Fluoro-2-nitrobenzoic acid |
| Molecular Structure | 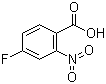 |
| Molecular Formula | C7H4FNO4 |
| Molecular Weight | 185.11 |
| CAS Registry Number | 394-01-4 |
| EC Number | 206-890-9 |
| SMILES | C1=CC(=C(C=C1F)[N+](=O)[O-])C(=O)O |
| Density | 1.6±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 140 - 145 °C (Expl.) |
| Boiling point | 343.8±27.0 °C 760 mmHg (Calc.)* |
| Flash point | 161.7±23.7 °C (Calc.)* |
| Index of refraction | 1.588 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
4-Fluoro-2-nitrobenzoic acid is an aromatic organic compound that belongs to the class of nitrobenzoic acids, specifically characterized by a fluorine atom substituted at the para position (position 4) and a nitro group at the ortho position (position 2) relative to the carboxylic acid group on the benzene ring. Its molecular formula is C7H4FNO4, and it exists as a solid under standard conditions. The synthesis of 4-fluoro-2-nitrobenzoic acid typically involves electrophilic aromatic substitution or nitration reactions of fluorobenzoic acid derivatives. One common method starts with 4-fluorobenzoic acid, which undergoes nitration using concentrated nitric and sulfuric acids. Due to the directing effects of the fluorine atom (which is ortho/para directing) and the carboxylic acid group (which is meta directing), a nitration reaction can be optimized to yield the 2-nitro derivative as a major product under controlled temperature and reaction time. This compound is a versatile intermediate in organic synthesis, especially in the production of pharmaceutical agents, agrochemicals, and advanced materials. Its structural features provide both electron-withdrawing and directing effects, which are useful for further chemical modifications, such as amide formation, esterification, or coupling reactions. The presence of both the nitro and carboxylic acid groups makes the molecule a good candidate for reactions involving nucleophilic aromatic substitution (especially at the position ortho or para to the fluorine atom), which is a valuable trait in medicinal chemistry. In medicinal chemistry, nitrobenzoic acid derivatives including 4-fluoro-2-nitrobenzoic acid have been investigated for their potential as anti-inflammatory, antimicrobial, and anticancer agents. The electron-deficient nature of the aromatic ring enhances its interaction with biological targets, particularly enzymes and receptors that are sensitive to electrostatic interactions. Furthermore, the fluorine atom can significantly impact the metabolic stability and membrane permeability of derived compounds. Due to the nitro group, 4-fluoro-2-nitrobenzoic acid can also serve as a precursor to anilines via catalytic or reducing processes such as hydrogenation or the use of reducing agents like iron in acidic media. The resulting fluoroaniline derivatives are important in the design of dyes, pigments, and bioactive molecules. In industrial and research laboratories, this compound is used as a starting material or building block in the synthesis of more complex aromatic systems. It is often employed in Suzuki or Heck coupling reactions following derivatization to suitable halides or esters. The carboxylic acid functionality can also be converted to acid chlorides, which are then used in acylation reactions to generate amides or esters. Overall, 4-fluoro-2-nitrobenzoic acid is valued for its reactivity and structural features that allow selective transformation in organic synthesis. Its role as a scaffold in medicinal and materials chemistry continues to support its use in both academic and industrial research. References 2024. Quinazolinone-based subchemotypes for targeting HIV-1 capsid protein: design and synthesis. Medicinal Chemistry Research, 33(10). DOI: 10.1007/s00044-024-03305-0 |
| Market Analysis Reports |