Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Shanghai ZaiQi Bio-Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemzq.com | |||
 | +86 (21) 5482-4098 | |||
 | +86 (21) 5482-4069 | |||
 | sales1@chemzq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Shanghai Yongzeng Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.allyzchem.com | |||
 | +86 (21) 6528-1118 | |||
 | +86 (21) 6528-2963 | |||
 | sales@yongzchem.com yongzchem@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Ricci Chimica | Italy | |||
|---|---|---|---|---|
 | www.riccichimica.com | |||
 | +39 (75) 692-9241 | |||
 | +39 (75) 592-6595 | |||
 | info@riccichimica.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Indoles |
|---|---|
| Name | 5-Fluoro-2-methylindole |
| Synonyms | 5-Fluoro-2-methyl-1H-indole |
| Molecular Structure | 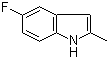 |
| Molecular Formula | C9H8FN |
| Molecular Weight | 149.16 |
| CAS Registry Number | 399-72-4 |
| EC Number | 609-764-5 |
| SMILES | CC1=CC2=C(N1)C=CC(=C2)F |
| Density | 1.2±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 98 - 101 °C (Expl.) |
| Boiling point | 269.2±20.0 °C 760 mmHg (Calc.)* |
| Flash point | 116.6±21.8 °C (Calc.)* |
| Index of refraction | 1.627 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
5‑Fluoro‑2‑methylindole is a fluorinated indole derivative with the molecular formula C9H8FN and CAS number 399‑72‑4. The indole nucleus (a benzene ring fused to a pyrrole) is substituted at the 5‑position with a fluorine atom, and at the 2‑position with a methyl group. These substituents confer both electronic and steric effects that can influence reactivity, biological activity, and interactions in synthetic applications. Because of the electron-withdrawing nature of the fluorine atom and the electron-donating effect of the methyl group, 5‑fluoro‑2‑methylindole can act as a useful building block in medicinal chemistry. The fluorine can enhance metabolic stability, influence lipophilicity, and potentially engage in hydrogen-bond modulation, while the methyl increases hydrophobic character and provides a site for further functionalization or modification. In synthetic chemistry, this compound is valuable for the preparation of more complex indole derivatives. The indole NH (pyrrole) can undergo acylation, alkylation, or metal-catalyzed cross-coupling reactions. The methyl group may serve as a handle for CH-activation chemistry, enabling functionalization at nearby positions. The overall indole scaffold, with its fluorine and methyl substituents, is often leveraged to create heterocycles, pharmacophores, and molecule libraries for drug discovery. From a practical standpoint, 5‑fluoro‑2‑methylindole is supplied as a solid and must be handled under inert atmosphere to avoid oxidation. According to available data, it has a melting point around 98–101 °C. Storage recommendations from catalog sources include protecting it from light and keeping it under an inert gas. Its solubility profile is typical for indoles of this type: it is soluble in common organic solvents such as methanol or dimethylformamide, but not highly soluble in aqueous media. References Using Phosphorus Pentasulfide (2019) Science of Synthesis DOI: 10.1055/s-0037-1612028 Palladium Complexes as Catalysts (2017) Science of Synthesis DOI: 10.1055/s-0037-1612028 C–N Coupling and Heck Coupling Reactions (2016) Science of Synthesis DOI: 10.1055/s-0037-1612028 |
| Market Analysis Reports |