Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Orgpharma Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.orgpharma.com | |||
 | +86 (21) 2206-2033 | |||
 | +86 (21) 5870-2436 | |||
 | info@orgpharma.cn sales@orgpharma.cn | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| DesignChem Pharmceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.designchem.com | |||
 | +86 (838) 606-5070 +86 18708362645 | |||
 | +86 (838) 823-1321 | |||
 | sales@designchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Qichuang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qc-chemical.com | |||
 | +86 (571) 8893-5129 | |||
 | +86 (571) 8825-0182 | |||
 | david@qc-chemical.com sales@qc-chemical.com davidw0828@gmail.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Renheng Pharm Limited | China | |||
|---|---|---|---|---|
 | www.renheng.com.cn | |||
 | +86 (20) 3998-1653 | |||
 | +86 (20) 3998-1158 | |||
 | sales@renheng.com.cn | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Enki Biopharmaceuticals (Shanghai) Limited | China | |||
|---|---|---|---|---|
 | www.enkibiopharma.com | |||
 | +86 (21) 5768-0965 +86 13916707528 | |||
 | +86 (21) 5768-0922 | |||
 | info@enkibiopharma.com | |||
 | QQ Chat | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shandong Chemed Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemedpharma.com | |||
 | +86 (531) 8880-3416 | |||
 | +86 (531) 8880-3416 | |||
 | sales@chemedpharma.com | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| R&D Scientific Inc. | Canada | |||
|---|---|---|---|---|
 | www.rdscientific.com | |||
 | +1 (226) 600-0236 | |||
 | sales@rdscientific.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Hangzhou Kehui Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hzkehuipharma.com | |||
 | +86 (0571) 8735-9773 | |||
 | tess5@tsbiopharm.com | |||
| Chemical distributor since 2017 | ||||
| chemBlink Standard supplier since 2023 | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Classification | API >> Synthetic anti-infective drugs >> Quinolone |
|---|---|
| Name | Besifloxacin hydrochloride |
| Synonyms | (R)-7-(3-Aminohexahydro-1H-azepin-1-yl)-8-chloro-1-cyclopropyl-6-fluoro-1,4-dihydro-4-oxo-3-quinolinecarboxylic acid hydrochloride |
| Molecular Structure | 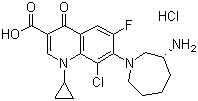 |
| Molecular Formula | C19H21ClFN3O3.HCl |
| Molecular Weight | 430.31 |
| CAS Registry Number | 405165-61-9 |
| EC Number | 696-612-6 |
| SMILES | C1CCN(C[C@@H](C1)N)C2=C(C=C3C(=C2Cl)N(C=C(C3=O)C(=O)O)C4CC4)F.Cl |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Besifloxacin hydrochloride, a fourth-generation fluoroquinolone antibiotic, was developed by SSP Co., Ltd. through extensive research aimed at improving the efficacy and safety profile of existing antibiotics. Chemists modified the fluoroquinolone scaffold to enhance its activity against a broad spectrum of bacteria while minimizing the risk of resistance development. Besifloxacin hydrochloride emerged from this endeavor as a potent antimicrobial agent with favorable pharmacokinetic properties. Its discovery marked a significant milestone in the fight against bacterial infections, particularly ocular infections caused by susceptible pathogens. Besifloxacin hydrochloride is primarily used to treat bacterial conjunctivitis, commonly known as pink eye. Its broad-spectrum activity against Gram-positive and Gram-negative bacteria, including resistant strains, makes it effective in resolving ocular infections. Clinical studies have demonstrated the efficacy and safety of besifloxacin hydrochloride in treating bacterial conjunctivitis, with rapid resolution of symptoms and low rates of adverse effects. Its favorable pharmacokinetic profile allows for convenient dosing regimens, making it suitable for both adults and children. Besifloxacin hydrochloride is also used in dermatology for the topical treatment of skin infections caused by susceptible bacteria. Its broad-spectrum activity and low potential for inducing resistance make it an effective option for managing bacterial skin conditions. In dermatological applications, besifloxacin hydrochloride has been shown to be well-tolerated with minimal systemic absorption, reducing the risk of systemic side effects commonly associated with oral antibiotics. This makes it suitable for long-term use in chronic skin conditions. As a fourth-generation fluoroquinolone, besifloxacin hydrochloride plays a role in antibiotic stewardship by offering an alternative treatment option with a lower risk of inducing bacterial resistance compared to older fluoroquinolones. Its use helps preserve the effectiveness of other antibiotics for more serious infections. Besifloxacin hydrochloride's unique chemical structure and mechanism of action minimize cross-resistance with other fluoroquinolones, making it valuable in the management of infections caused by multidrug-resistant bacteria. Besifloxacin hydrochloride's discovery and development underscore the importance of continuous research and innovation in antibiotic therapy. Its success highlights the potential for designing novel antimicrobial agents with improved efficacy, safety, and resistance profiles through rational drug design. Ongoing research aims to explore additional therapeutic applications of besifloxacin hydrochloride, including its potential in combination therapies and as a prophylactic agent for preventing surgical site infections and bacterial keratitis. References 2018. Besifloxacin: A Critical Review of Its Characteristics, Properties, and Analytical Methods. Critical Reviews in Analytical Chemistry, 48(6). DOI: 10.1080/10408347.2018.1429885 2015. Besifloxacin Ophthalmic Suspension 0.6% in the Treatment of Bacterial Keratitis: A Retrospective Safety Surveillance Study. Journal of Ocular Pharmacology and Therapeutics, 31(3). DOI: 10.1089/jop.2014.0039 2009. Target specificity of the new fluoroquinolone besifloxacin in Streptococcus pneumoniae, Staphylococcus aureus and Escherichia coli. Journal of Antimicrobial Chemotherapy, 63(3). DOI: 10.1093/jac/dkn528 |
| Market Analysis Reports |