Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Sichuan Guanghan Bio-Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sino-produce.com | |||
 | +86 (28) 8612-7071 | |||
 | +86 (28) 8612-7072 | |||
 | sales@sino-produce.com | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Hubei Research Institute of Chemistry | China | |||
|---|---|---|---|---|
 | www.haisopharm.com | |||
 | +86 (27) 8742-2225 | |||
 | +86 (27) 8749-6702 | |||
 | info@haisopharm.com | |||
| Chemical manufacturer since 1973 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Sichuan Weibo Science and Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.scweibo.com | |||
 | +86 (28) 8757-8473 +86 13880582998 | |||
 | +86 (28) 8757-8473 | |||
 | dudes@163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Jinan Zhongke Yitong Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jnzkyt.com | |||
 | +86 (531) 8865-8009 | |||
 | +86 (531) 8895-6078 | |||
 | 2871367557@qq.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Zhejiang Synose Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.synose.com | |||
 | +86(579)8227-5537 8917-2081 | |||
 | +86 (579) 8227-7133 | |||
 | expo@synose.com | |||
| Chemical manufacturer since 1996 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Molcan Corporation | USA | |||
|---|---|---|---|---|
 | www.molcan.com | |||
 | +1 (905) 731-5537 | |||
 | +1 (905) 248-3843 | |||
 | info@molcan.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Shanghai Hongbang Medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hongbangpharma.com | |||
 | +86 (21) 2428-0809 +86 13671516988 | |||
 | +86 (21) 2428-0809 | |||
 | kimberly-hongbangpharma@hotmail.com 1377976036@qq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Xinxiang Aurora Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aurora-biotech.com | |||
 | +86 (373) 308-8722 +86 18637352520 | |||
 | +86 (373) 308-8722 | |||
 | info@aurora-biotech.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Indofine Chemical Company, Inc. | USA | |||
|---|---|---|---|---|
 | www.indofinechemical.com | |||
 | +1 (888) 463-6346 | |||
 | +1 (908) 359-1179 | |||
 | info@indofinechemical.com | |||
| Chemical manufacturer since 1981 | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Orgentis Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.orgentis.com | |||
 | +49 39482-355 | |||
 | +49 39482-79071 | |||
 | sales@orgentis.com | |||
| Chemical manufacturer since 1991 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyran compound |
|---|---|
| Name | 2,3,4,6-Tetra-O-benzyl-D-glucopyranose |
| Synonyms | (3R,4S,5R,6R)-3,4,5-tris(phenylmethoxy)-6-(phenylmethoxymethyl)oxan-2-ol |
| Molecular Structure | 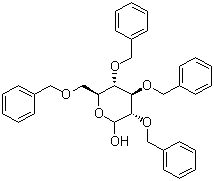 |
| Molecular Formula | C34H36O6 |
| Molecular Weight | 540.66 |
| CAS Registry Number | 4132-28-9 |
| EC Number | 609-908-7 |
| SMILES | C1=CC=C(C=C1)COC[C@@H]2[C@H]([C@@H]([C@H](C(O2)O)OCC3=CC=CC=C3)OCC4=CC=CC=C4)OCC5=CC=CC=C5 |
| Density | 1.2±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 151-156 °C (Expl.) |
| Index of Refraction | 1.619, Calc.* |
| Boiling Point | 672.4±55.0 °C (760 mmHg), Calc.* |
| Flash Point | 360.4±31.5 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
2,3,4,6-Tetra-O-benzyl-D-glucopyranose is a glucopyranose derivative where all the hydroxyl groups on the glucose molecule are protected with benzyl groups. This compound is an important intermediate in carbohydrate chemistry, widely used for synthesizing other complex carbohydrate derivatives. The benzyl groups act as protecting groups, preventing unwanted reactions during further chemical manipulations and enabling selective functionalization of the glucose molecule. The discovery of 2,3,4,6-Tetra-O-benzyl-D-glucopyranose can be traced back to the development of protecting group strategies in carbohydrate chemistry. The protection of hydroxyl groups using benzyl groups is a well-established method that allows for the selective derivatization of other positions on the glucose molecule. This strategy has proven essential in the synthesis of oligosaccharides, glycoconjugates, and other carbohydrate-based compounds, which have significant applications in both pharmaceuticals and materials science. The synthesis of 2,3,4,6-Tetra-O-benzyl-D-glucopyranose typically involves the selective benzylation of D-glucopyranose in the presence of a base, such as potassium carbonate or sodium hydride. The reaction is carried out in an appropriate solvent, often anhydrous dimethylformamide (DMF), which helps facilitate the formation of the benzyl ether groups. This reaction selectively protects the hydroxyl groups at the 2, 3, 4, and 6 positions, leaving the glucose molecule in its protected form. Once the desired synthesis is complete, the benzyl groups can be selectively removed in subsequent steps to expose the hydroxyl groups for further reactions, such as glycosylation or coupling with other biomolecules. The applications of 2,3,4,6-Tetra-O-benzyl-D-glucopyranose are vast and varied. In the field of carbohydrate chemistry, it serves as a valuable intermediate for the synthesis of complex oligosaccharides, which are important for studying glycobiology, the interaction between carbohydrates and proteins. These compounds are often used in drug discovery, as carbohydrates play a critical role in cellular recognition and signaling processes. Moreover, 2,3,4,6-Tetra-O-benzyl-D-glucopyranose is frequently employed in the development of glycoproteins, which are used as therapeutic agents for diseases like cancer and viral infections. In the realm of materials science, 2,3,4,6-Tetra-O-benzyl-D-glucopyranose is used to modify and functionalize surfaces and polymers. Its ability to introduce hydroxyl groups at specific positions on the glucose ring allows for the design of carbohydrate-based materials with tailored properties. These materials are often employed in creating biocompatible surfaces, sensors, and bioactive materials for medical applications. Furthermore, the versatility of this compound in terms of its protection and deprotection chemistry makes it an essential tool for developing new synthetic routes and materials. In conclusion, 2,3,4,6-Tetra-O-benzyl-D-glucopyranose is a crucial intermediate in carbohydrate chemistry, enabling the synthesis of complex sugar derivatives for use in a wide range of applications. Its ability to serve as a precursor for oligosaccharides and glycoconjugates underscores its importance in pharmaceutical and materials science. With continued research and development, 2,3,4,6-Tetra-O-benzyl-D-glucopyranose is likely to play an even larger role in advancing the understanding of carbohydrate-related processes and developing new therapeutic materials. References 2016. A Convenient, Gram-Scale Synthesis of 1-Deoxymannojirimycin. Synthesis, 48(06). DOI: 10.1055/s-0035-1561323 2008. Stereoselective Synthesis of Branched Cyclopentitols by Titanium(III)-Promoted Reductive Cyclization of 4-Oxiranylaldehydes and 4-Oxiranyl Ketones Derived from Hexoses. Synthesis, 2008(18). DOI: 10.1055/s-2008-1067257 2002. Potential bile acid metabolites. 24. An efficient synthesis of carboxyl-linked glucosides and their chemical properties. Lipids, 37(1). DOI: 10.1007/s11745-002-0869-3 |
| Market Analysis Reports |