Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Shaanxi Kaida Chemcial Engineering Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kd-chem.com | |||
 | +86 (917) 331-7143 332-2918 | |||
 | +86 (917) 331-0616 | |||
 | kaida@kd-chem.com | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2014 | ||||
| W.C.Heraeus GmbH | Germany | |||
|---|---|---|---|---|
 | www.heraeus-chemicalproducts.com | |||
 | +49 (6181) 355-707 | |||
 | +49 (6181) 355-914 | |||
 | info@heraeus.com | |||
| Chemical manufacturer | ||||
| Heraeus Ltd. | Hong Kong | |||
|---|---|---|---|---|
 | www.heraeus.hk | |||
 | +852 2675-1200 2773-1733 | |||
 | +852 2773-1090 / 2682-3220 | |||
 | cpd@heraeus.com.hk | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Colonial Metals, Inc. | USA | |||
|---|---|---|---|---|
 | www.colonialmetals.com | |||
 | +1 (410) 398-7200 (800) 962-1537 | |||
 | +1 (410) 398-2918 | |||
 | sales@colonialmetals.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic rhodium |
|---|---|
| Name | Rhodium acetate |
| Synonyms | Acetoxyrhodium |
| Molecular Structure | 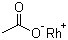 |
| Molecular Formula | C2H3O2Rh |
| Molecular Weight | 161.95 |
| CAS Registry Number | 42204-14-8 |
| EC Number | 255-707-9 |
| SMILES | [Rh].[O-]C(=O)C |
| Melting point | 238 °C (decomp) (Expl.) |
|---|---|
| Hazard Symbols |   GHS07;GHS08 Warning Details GHS07;GHS08 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H341-H319 Details | ||||||||||||||||||||
| Safety Statements | P280-P281-P305+P351+P338-P308+P313-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Rhodium acetate is a coordination compound of rhodium with the general formula Rh2(OAc)4, where OAc represents the acetate group (CH3COO−). More formally known as dirhodium tetraacetate, this compound consists of a dimeric structure in which two rhodium atoms are bridged by four acetate ligands. It is a well-established organometallic complex that has found broad use in homogeneous catalysis and synthetic organic chemistry. The compound was first described in the mid-20th century during investigations into the coordination chemistry of rhodium carboxylates. Its synthesis typically involves reacting rhodium(III) chloride hydrate with sodium acetate or acetic acid under reflux conditions in aqueous or alcoholic solutions. The resulting compound is isolated as a deep green crystalline solid that is stable under ambient conditions and moderately soluble in organic solvents such as methanol, acetone, and dichloromethane. Structurally, dirhodium tetraacetate is characterized by a direct metal–metal bond between the two rhodium atoms, with a formal bond order of one. Each rhodium atom is further coordinated by four oxygen atoms from the acetate ligands, giving each metal center a square pyramidal geometry. This bimetallic structure imparts unique electronic properties that are central to its reactivity and utility in catalysis. One of the primary applications of rhodium acetate is as a catalyst in carbene transfer reactions, particularly in metal-catalyzed C–H insertion and cyclopropanation processes. In these reactions, the compound activates diazo compounds to generate reactive rhodium-carbenoid intermediates. These intermediates can then insert into C–H bonds or react with alkenes to form cyclopropanes with high regio- and stereoselectivity. This transformation is widely used in the synthesis of natural products and pharmaceuticals, as it enables the construction of complex carbon frameworks under mild conditions. Rhodium acetate is also employed in the oxidation of alcohols and olefins, typically in conjunction with co-oxidants such as tert-butyl hydroperoxide. In addition, it has been used in aziridination and nitrene transfer reactions, where it facilitates the formation of three-membered N-containing heterocycles from iminoiodinanes or similar precursors. In the field of materials chemistry, dirhodium tetraacetate has been investigated for its magnetic and electronic properties due to the presence of a direct metal–metal bond. Its bimetallic nature also serves as a model for studying metal–metal interactions in bioinorganic and solid-state chemistry. Analytically, rhodium acetate is characterized using techniques such as X-ray crystallography, which reveals its paddlewheel-like dimeric structure, and nuclear magnetic resonance (NMR) spectroscopy, which confirms the presence of symmetric acetate ligands. Ultraviolet-visible (UV-Vis) spectroscopy and cyclic voltammetry provide further insight into its electronic transitions and redox properties. The compound is generally stable in air and light but should be handled with care due to the potential toxicity of rhodium and its compounds. It is used in catalytic amounts, and proper precautions should be taken during its handling and disposal. Its cost and limited availability compared to other transition metal catalysts may restrict its use to specialized applications where its unique reactivity is essential. In summary, rhodium acetate is a well-characterized dirhodium(II) complex with a wide range of catalytic applications. Its stable bimetallic core and versatile ligand environment enable highly selective transformations in organic synthesis, particularly in carbene transfer and C–H functionalization chemistry. Its structure and reactivity have made it a valuable tool in both academic research and industrial applications. References none |
| Market Analysis Reports |