Online Database of Chemicals from Around the World
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Quinoline compound |
|---|---|
| Name | NNMTi |
| Synonyms | 5-Amino-1-methylquinolinium iodide |
| Molecular Structure | 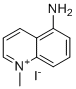 |
| Molecular Formula | C10H11IN2 |
| Molecular Weight | 286.11 |
| CAS Registry Number | 42464-96-0 |
| SMILES | C[N+]1=CC=CC2=C(C=CC=C21)N.[I-] |
| Solubility | soluble 100 mM (DMSO) (Expl.) |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P312-P330-P332+P313-P337+P313-P362-P403+P233-P405-P501 Details |
|
Nicotinamide N-methyltransferase Inhibitors (NNMTi) are a class of chemical compounds developed to inhibit the activity of nicotinamide N-methyltransferase (NNMT), a cytosolic enzyme responsible for methylating nicotinamide into 1-methylnicotinamide using S-adenosylmethionine as a methyl donor. NNMT is implicated in various metabolic and pathological processes, including obesity, cancer, diabetes, and neurological disorders, making its inhibitors critical for therapeutic development. The discovery of NNMTi stems from efforts to target dysregulated metabolism and aberrant methylation pathways. Elevated NNMT activity has been observed in numerous cancers, including breast, pancreatic, and lung cancers, where it contributes to tumor growth and resistance to apoptosis. Similarly, NNMT overexpression in adipose tissue has been linked to obesity-related insulin resistance. Recognizing these associations, researchers began developing NNMT inhibitors to restore normal metabolic balance and mitigate disease progression. NNMTi compounds are designed with high specificity to inhibit the catalytic activity of NNMT. Structural studies of NNMT have revealed key binding pockets that accommodate nicotinamide and S-adenosylmethionine. Early inhibitors were small molecules that mimicked these substrates, competing with them to block the enzyme's activity. Advances in medicinal chemistry have since yielded more potent and selective inhibitors with improved pharmacokinetics and reduced off-target effects. The therapeutic applications of NNMTi are expanding rapidly. In oncology, NNMT inhibitors have demonstrated potential in preclinical studies to suppress tumor proliferation and enhance the efficacy of existing chemotherapeutics. By disrupting the NNMT-mediated methylation pathway, these inhibitors impair the energy metabolism and epigenetic adaptations of cancer cells. In metabolic disorders, NNMTi compounds are being investigated for their ability to improve insulin sensitivity and promote weight loss, addressing the root causes of type 2 diabetes and obesity. Furthermore, NNMT inhibitors are emerging as promising candidates in neurodegenerative disease research. NNMT has been implicated in the pathogenesis of Alzheimer's and Parkinson's diseases through its effects on cellular methylation states and oxidative stress. By modulating NNMT activity, researchers hope to develop novel interventions that slow neurodegeneration and enhance neuronal resilience. The development of NNMT inhibitors has also spurred interest in diagnostic applications. Elevated NNMT levels in biological fluids are considered potential biomarkers for cancer and metabolic disorders. NNMTi compounds could thus play a role in both therapeutic and diagnostic strategies, supporting a more personalized approach to treatment. Ongoing research aims to optimize NNMT inhibitors for clinical use. Challenges such as off-target interactions, bioavailability, and long-term safety are being addressed through rational drug design and advanced screening methodologies. As the understanding of NNMT’s role in health and disease continues to grow, NNMTi compounds are poised to become valuable tools in precision medicine. References 1976. [Antibacterial activity of sisomicin in comparison with gentamicin]. Arzneimittel-Forschung, 26(5). URL: https://pubmed.ncbi.nlm.nih.gov/9099 2017. Structure-Activity Relationship for Small Molecule Inhibitors of Nicotinamide N-Methyltransferase. Journal of Medicinal Chemistry, 60(12). DOI: 10.1021/acs.jmedchem.7b00389 2024. Control of NAD+ homeostasis by autophagic flux modulates mitochondrial and cardiac function. The EMBO Journal, 43(4). DOI: 10.1038/s44318-023-00009-w |
| Market Analysis Reports |