Online Database of Chemicals from Around the World
| Zhejiang Chempharm Industry&trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chempharm.cn | |||
 | +86 (571) 8770-1568 | |||
 | export@chempharm.cn | |||
| Chemical distributor since 1996 | ||||
| chemBlink Standard supplier since 2007 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Vanz Pharm Inc. | China | |||
|---|---|---|---|---|
 | www.vanzpharm.com | |||
 | +86 (27) 8425-4881 | |||
 | +86 (27) 8425-4882 | |||
 | 1009531590@qq.com | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Wuhan Hengheda Pharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hhdpharm.com | |||
 | +86 18086046872 | |||
 | hhdpharm@hotmail.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Classification | Biochemical >> Amino acids and their derivatives >> Proline derivatives |
|---|---|
| Name | trans-3-Hydroxy-L-proline |
| Synonyms | (2S,3S)-(-)-3-Hydroxy-2-pyrrolinecarboxylic acid |
| Molecular Structure | 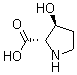 |
| Molecular Formula | C5H9NO3 |
| Molecular Weight | 131.13 |
| CAS Registry Number | 4298-08-2 |
| EC Number | 639-470-2 |
| SMILES | C1CN[C@@H]([C@H]1O)C(=O)O |
| Density | 1.395 |
|---|---|
| Melting point | 235 °C |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
Trans-3-Hydroxy-L-proline is a chiral amino acid derivative with a unique hydroxyl group attached to the proline ring in a trans configuration. Due to its unique structural features, this compound has had a significant impact in various fields of science and industry. Trans-3-Hydroxy-L-proline was first synthesized in the mid-20th century as part of research into modified proline derivatives. Scientists were interested in understanding how substituents, such as the hydroxyl group, affect the properties of proline and its analogs. The introduction of a hydroxyl group at the 3-position of the proline ring while maintaining the trans configuration resulted in this compound. Its synthesis provides insights into the role of hydroxylation in altering the chemical and biological behavior of proline. In drug development, trans-3-Hydroxy-L-proline is explored for its potential therapeutic properties. Its unique structure can influence the activity of pharmaceutical agents, making it useful as a chiral building block for designing new drugs. Researchers have studied its role in creating compounds with specific biological effects, such as anti-inflammatory or antiviral agents. This compound is used as an intermediate in organic synthesis. Its hydroxyl group and chiral center enable the creation of complex molecules with desired properties. It is used in the development of fine chemicals, agrochemicals, and other organic compounds, using its reactivity to facilitate various chemical transformations. In biochemical research, trans-3-hydroxy-L-proline is used to study the effects of hydroxylation on protein structure and function. Its effects on protein folding and stability provide valuable insights into the role of modified proline residues in biological systems, contributing to a better understanding of protein chemistry. The properties of this compound are also explored in materials science to develop advanced materials. Its unique structure can be used to create materials with specific mechanical or electronic properties, thereby promoting innovation in technology and materials science. References 1962. Hydroxyproline in the developing chick embryo. The Journal of Biological Chemistry, 237(7). DOI: 10.1016/s0021-9258(19)63379-6 2023. Structures of L-proline trans-hydroxylase reveal the catalytic specificity and provide deeper insight into AKG-dependent hydroxylation. Acta Crystallographica Section D Structural Biology, 79(Pt 4). DOI: 10.1107/s2059798323001936 |
| Market Analysis Reports |