Online Database of Chemicals from Around the World
| Shanghai RC Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rcc.net.cn | |||
 | +86 (21) 5032-2175 5866-1250 ex 603 | |||
 | +86 (21) 5032-2176 / 5866-1251 | |||
 | sales@rcc.net.cn chad@rcc.net.cn | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Jiangxi Time Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.groupchem.com | |||
 | +86 (794) 718-3888 718-3887 526-9970 +86 13867635890 | |||
 | +86 (794) 526-9668 / 526-9166 | |||
 | sales@groupchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2011 | ||||
| SynQuest Labs, Inc. | USA | |||
|---|---|---|---|---|
 | www.synquestlabs.com | |||
 | +1 (386) 462-0788 | |||
 | +1 (386) 462-7097 | |||
 | Sales@synquestlabs.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Xinlian Rui Biomedical Technology (shandong) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xlrchem.com | |||
 | +86 15153173989 | |||
 | 3865799121@qq.com | |||
 | QQ Chat | |||
 | WeChat: 15153173989 | |||
| Chemical manufacturer since 2024 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| P and M Invest Ltd. | Russia | |||
|---|---|---|---|---|
 | www.fluorine.ru | |||
 | +7 (495) 135-6494 | |||
 | +7 (495) 135-6509 | |||
 | sales@fluorine1.ru | |||
| Chemical manufacturer | ||||
| City Chemical LLC | USA | |||
|---|---|---|---|---|
 | www.citychemical.com | |||
 | +1 (203) 932-2489 | |||
 | +1 (203) 937-8400 | |||
 | sales@citychemical.com | |||
| Chemical distributor | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Sinbond Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinbondpharm.com | |||
 | +86 (531) 8703-8285 +86 13583181986 +44 (208) 242-5518 | |||
 | +86 (531) 8703-8285 | |||
 | cici1124@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| Ricci Chimica | Italy | |||
|---|---|---|---|---|
 | www.riccichimica.com | |||
 | +39 (75) 692-9241 | |||
 | +39 (75) 592-6595 | |||
 | info@riccichimica.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Fatty acid |
|---|---|
| Name | Methyl difluoroacetate |
| Synonyms | methyl 2,2-difluoroacetate |
| Molecular Structure | 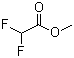 |
| Molecular Formula | C3H4F2O2 |
| Molecular Weight | 110.06 |
| CAS Registry Number | 433-53-4 |
| EC Number | 207-089-7 |
| SMILES | COC(=O)C(F)F |
| Density | 1.2±0.1 g/cm3 Calc.*, 1.272 g/mL (Expl.) |
|---|---|
| Boiling point | 45.0±25.0 °C 760 mmHg (Calc.)*, 85 - 86 °C (Expl.) |
| Flash point | -16.9±18.1 °C (Calc.)*, 19 °C (Expl.) |
| Index of refraction | 1.312 (Calc.)*, 1.334 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS02;GHS05;GHS07 Danger Details GHS02;GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H226-H302-H312-H314-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P260-P261-P264-P264+P265-P270-P271-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P303+P361+P353-P304+P340-P305+P351+P338-P305+P354+P338-P316-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P363-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1993 | ||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
Methyl difluoroacetate is a fluorinated ester with the molecular formula CHF2COOCH3. It is the methyl ester of difluoroacetic acid and belongs to the class of organofluorine compounds that have been studied for their unique electronic and chemical properties. The two fluorine atoms attached to the alpha carbon are strongly electron-withdrawing, increasing the electrophilicity of the carbonyl carbon and influencing the reactivity of the ester. Methyl difluoroacetate is typically a colorless liquid, soluble in organic solvents, and reactive under nucleophilic substitution or hydrolysis conditions. The primary application of methyl difluoroacetate is as a building block in organic synthesis. It serves as a source of the difluoroacetyl group for introducing difluoromethyl functionalities into target molecules. The compound undergoes nucleophilic acyl substitution reactions with alcohols, amines, and thiols to form esters, amides, and thioesters, which are valuable intermediates for pharmaceuticals, agrochemicals, and specialty chemicals. The electron-withdrawing effects of the difluoromethyl group enhance chemical stability and modulate the reactivity of downstream products, making it an important reagent for synthetic chemists. In medicinal chemistry, methyl difluoroacetate is used to synthesize difluoromethylated drug candidates. The difluoromethyl group can act as a bioisostere for methyl or hydroxyl groups, improving metabolic stability, lipophilicity, and pharmacokinetic properties of the resulting molecules. It has been applied in the preparation of enzyme inhibitors, antiviral agents, and other bioactive compounds, where incorporation of the difluoromethyl group can enhance potency and chemical robustness. The ester functionality allows controlled reactions under mild conditions, enabling selective functionalization of sensitive intermediates. In agrochemical research, methyl difluoroacetate is used to prepare fluorinated herbicides, fungicides, and insecticides. Introduction of the difluoromethyl group into precursor molecules increases chemical and thermal stability, lipophilicity, and biological activity. Its reactivity as a methyl ester allows efficient acyl transfer, facilitating the synthesis of bioactive intermediates that can be further transformed into final agrochemical products. The compound also finds application in materials science and specialty chemical synthesis. Fluorinated esters derived from methyl difluoroacetate are employed to introduce difluoromethyl groups into polymers, coatings, and functionalized surfaces, enhancing hydrophobicity, chemical resistance, and thermal stability. The ester’s reactivity enables selective incorporation into complex molecules, contributing to improved material properties. The discovery and use of methyl difluoroacetate underscore its importance as a versatile fluorinated reagent. Its combination of reactivity, electron-withdrawing effects, and chemical stability makes it a valuable intermediate in modern organic synthesis, supporting applications in pharmaceuticals, agrochemicals, materials science, and specialty chemicals. The compound exemplifies how incorporation of fluorinated functional groups can enhance the utility of simple esters in research and industrial applications. References 2023. Fluorination in advanced battery design. Nature Reviews Materials, 9(1). DOI: 10.1038/s41578-023-00623-4 2014. Theoretical investigation of the atmospheric chemistry of methyl difluoroacetate: reaction with Cl atoms and fate of alkoxy radical at 298 K. Structural Chemistry, 25(2). DOI: 10.1007/s11224-014-0425-3 2016. Efficient synthesis of tri- and difluoroacetyl hydrazides as useful building blocks for non-symmetrically substituted, fluoroalkylated 1,3,4-oxadiazoles. Chemistry of Heterocyclic Compounds, 52(2). DOI: 10.1007/s10593-016-1845-3 |
| Market Analysis Reports |