Online Database of Chemicals from Around the World
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Extrasynthese Chemical S.A.S. | France | |||
|---|---|---|---|---|
 | www.extrasynthese.com | |||
 | +33 (47) 898-2034 | |||
 | +33 (47) 898-1945 | |||
 | info@extrasynthese.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Shaanxi Yongyuan Bio-Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.herbalextractcn.com | |||
 | +86 (29) 8834-6470 | |||
 | +86 (29) 8834-6470 | |||
 | infoyy@herbalextractcn.com yysale2@herbalextractcn.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Watec Laboratories, Inc. | China | |||
|---|---|---|---|---|
 | www.wateclaboratories.com | |||
 | +86 (519) 8692-1516 +86 18602586511 | |||
 | +86 (519) 8526-7382 | |||
 | info@wateclaboratories.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Xi'an Lvdong Science & Technology Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lvdost.cn | |||
 | +86 (29) 6232-2608 | |||
 | +86 (29) 6232-2608 | |||
 | lvdost@163.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Chengdu Biopurify Phytochemicals Ltd. | China | |||
|---|---|---|---|---|
 | www.phytopurify.com | |||
 | +86 (28) 8263-3860 8263-3987 | |||
 | +86 (28) 8263-3165 | |||
 | sales@biopurify.com biopurify@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Cfm Oskar Tropitzsch GmbH | Germany | |||
|---|---|---|---|---|
 | www.cfmot.de | |||
 | +49 (9231) 9619-0 | |||
 | +49 (9231) 9619-60 | |||
 | info@cfmot.de | |||
| Chemical distributor since 1985 | ||||
| chemBlink Standard supplier since 2022 | ||||
| R&D Scientific Inc. | Canada | |||
|---|---|---|---|---|
 | www.rdscientific.com | |||
 | +1 (226) 600-0236 | |||
 | sales@rdscientific.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Alfa Aesar. | USA | |||
|---|---|---|---|---|
 | www.alfa.com | |||
 | +1 (978) 521-6300 | |||
 | +1 (978) 521-6350 | |||
 | info@alfa.com | |||
| Chemical manufacturer | ||||
| City Chemical LLC | USA | |||
|---|---|---|---|---|
 | www.citychemical.com | |||
 | +1 (203) 932-2489 | |||
 | +1 (203) 937-8400 | |||
 | sales@citychemical.com | |||
| Chemical distributor | ||||
| Indofine Chemical Company, Inc. | USA | |||
|---|---|---|---|---|
 | www.indofinechemical.com | |||
 | +1 (888) 463-6346 | |||
 | +1 (908) 359-1179 | |||
 | info@indofinechemical.com | |||
| Chemical manufacturer since 1981 | ||||
| Cayman Chemical Company | USA | |||
|---|---|---|---|---|
 | www.caymanchem.com | |||
 | +1 (734) 971-3335 | |||
 | +1 (734) 971-3640 | |||
 | sales@caymanchem.com | |||
| Chemical manufacturer | ||||
| Ascent Scientific | UK | |||
|---|---|---|---|---|
 | www.ascentscientific.com | |||
 | +44 (117) 982-9988 | |||
 | +44 (2030) 700-369 | |||
 | customerservice@ascentscientific.co.uk | |||
| Chemical manufacturer | ||||
| ABX Advanced Biochemical Compounds | Germany | |||
|---|---|---|---|---|
 | www.abx.de | |||
 | +49 (3528) 4041-60 | |||
 | +49 (3528) 4041-65 | |||
 | info@abx.de | |||
| Chemical manufacturer | ||||
| VDM Biochemicals | USA | |||
|---|---|---|---|---|
 | www.vdmbio.com | |||
 | +1 (330) 252-8181 | |||
 | +1 (330) 252-8171 | |||
 | sales@vdmbio.com info@vdmbio.com | |||
| Chemical manufacturer | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Classification | Natural product >> Alkaloid |
|---|---|
| Name | Harmine |
| Synonyms | 7-Methoxy-1-methyl-9H-pyrido[3,4-b]indole; 7-Methoxy-1-methyl-9H-beta-carboline |
| Molecular Structure | 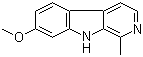 |
| Molecular Formula | C13H12N2O |
| Molecular Weight | 212.25 |
| CAS Registry Number | 442-51-3 |
| EC Number | 207-131-4 |
| SMILES | CC1=NC=CC2=C1NC3=C2C=CC(=C3)OC |
| Solubility | 100 mM (DMSO), 5 mM (ethanol) |
|---|---|
| Melting point | 262-264 °C |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P321-P330-P332+P317-P337+P317-P362+P364-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
Harmin is a naturally occurring beta-carboline alkaloid with the molecular formula C13H12N2O. It is best known for being found in the seeds of Peganum harmala (also known as Syrian rue), and in various species of the Banisteriopsis vine, the latter being the main ingredient in Ayahuasca, a psychoactive beverage used by indigenous Amazonian tribes. Harmin’s discovery dates back to the early 20th century, although its use in traditional medicine and shamanic rituals is much older. In 1847, French chemist Jean-François Persoz isolated harmala from Peganum harmala seeds. Further research into its structure and properties in the early 20th century led to a better understanding of its chemical properties and potential applications. Harmin has a rich history of ethnobotany and medicinal use. It has traditionally been used for its psychoactive effects, particularly in Ayahuasca rituals. This potion, a blend of Banisteriopsis caapi (rich in hamiemin) and Psychotria viridis (containing DMT), has been used for centuries by indigenous tribes in the Amazon basin for its purported psychotropic and healing properties. Hamiemin, along with the other beta-carbolines in the potion, acts as a monoamine oxidase inhibitor (MAOI), preventing the breakdown of DMT, thereby prolonging and enhancing its psychoactive effects. In modern medicine, hamiemin has attracted a great deal of interest for its pharmacological properties. Its primary mechanism of action as a MAOI makes it a candidate for treating conditions such as depression and anxiety. By inhibiting the breakdown of neurotransmitters such as serotonin and dopamine, hamiemin can potentially elevate mood and improve emotional well-being. Research into the antidepressant effects of hamiemin is ongoing, with some studies suggesting that it may be more beneficial than traditional antidepressants, particularly due to its rapid onset of action. In addition to its psychoactive and antidepressant properties, hamiemin has shown promise in a variety of biomedical applications. One area of interest is its potential anticancer properties. Research has shown that harmine can induce apoptosis (programmed cell death) in cancer cells and inhibit tumor cell proliferation. This makes it an interesting subject for investigation in the development of new cancer therapies. Harmine also has anti-inflammatory and neuroprotective properties. Research has shown that it can protect neurons from damage caused by oxidative stress and inflammation, which are underlying factors in neurodegenerative diseases such as Alzheimer's and Parkinson's. These properties make harmine a potential therapeutic agent to slow the progression of these debilitating diseases. In addition to its medicinal applications, harmine is also used in biochemical research. Its ability to insert into DNA and inhibit various enzymes makes it a valuable tool for studying cellular processes and enzyme function. This deepens our understanding of cell biology and has potential implications for the development of new drugs and therapies. The use of harmine is not without its challenges. Its psychoactive effects and potential for interactions with other drugs require careful management in the clinical setting. As with any potent bioactive compound, the dosage and method of administration need to be carefully controlled to maximize the therapeutic effect while minimizing side effects. References 1979. Biliary and urinary excretion of drug conjugates: Effect of diuresis and choleresis on excretion of harmol sulphate and harmol glucuronide in the rat. Xenobiotica, 9(1). DOI: 10.3109/00498257909038727 1991. [3H]Harman Binding Experiments. I: A Reversible and Selective Radioligand for Monoamine Oxidase Subtype A in the CNS of the Rat. Journal of Neurochemistry, 56(2). DOI: 10.1111/j.1471-4159.1991.tb08177.x 2021. Solid-Contact Potentiometric Sensors Based on Main-Tailored Bio-Mimics for Trace Detection of Harmine Hallucinogen in Urine Specimens. Molecules (Basel, Switzerland), 26(2). DOI: 10.3390/molecules26020324 |
| Market Analysis Reports |