Online Database of Chemicals from Around the World
| Extrasynthese Chemical S.A.S. | France | |||
|---|---|---|---|---|
 | www.extrasynthese.com | |||
 | +33 (47) 898-2034 | |||
 | +33 (47) 898-1945 | |||
 | info@extrasynthese.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| NetQem, LLC | USA | |||
|---|---|---|---|---|
 | www.netqem.us | |||
 | +1 (919) 544-4122 | |||
 | +1 (919) 544-4109 | |||
 | sales@netqem.us | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Labseeker Inc | USA | |||
|---|---|---|---|---|
 | www.labseeker.com | |||
 | +1 (858) 750-1632 | |||
 | +1 (858) 412-1220 | |||
 | marketing@labseeker.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Chengdu Biopurify Phytochemicals Ltd. | China | |||
|---|---|---|---|---|
 | www.phytopurify.com | |||
 | +86 (28) 8263-3860 8263-3987 | |||
 | +86 (28) 8263-3165 | |||
 | sales@biopurify.com biopurify@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2017 | ||||
| Jiangxi Cedar Natural Medicinal Oil Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jxxs8-1.com | |||
 | +86 18000738807 | |||
 | 2134411736@qq.com | |||
 | QQ Chat | |||
 | WeChat: 592432494 | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Food additive >> Food spice >> Natural equivalent spice |
|---|---|
| Name | (+)-Borneol |
| Synonyms | (1R,2S,4R)-Borneol; endo-(1R)-1,7,7-Trimethylbicyclo[2.2.1]heptan-2-ol |
| Molecular Structure | 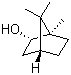 |
| Molecular Formula | C10H18O |
| Molecular Weight | 154.25 |
| CAS Registry Number | 464-43-7 |
| EC Number | 207-352-6 |
| FEMA | 2157 |
| SMILES | C[C@@]12CC[C@@H](C1(C)C)C[C@@H]2O |
| Density | 1.0±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 207 °C (Expl.) |
| Index of Refraction | 1.502, Calc.* |
| alpha | 36 ° (c=5 in EtOH) |
| Boiling Point | 212.0 °C (760 mmHg), Calc.* |
| Flash Point | 80.7±10.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS02 Danger Details GHS02 Danger Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H228 Details | ||||||||
| Safety Statements | P210-P240-P241-P280-P370+P378 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| Transport Information | UN 1312 | ||||||||
| SDS | Available | ||||||||
|
(+)-Borneol is a naturally occurring bicyclic monoterpene alcohol that is widely found in essential oils, particularly in species of the *Artemisia*, *Mentha*, and *Cinnamomum* genera. It has been utilized for centuries in traditional medicine and is also known for its distinct aroma. The compound is optically active, with the (+)-isomer being the most common and biologically relevant form. The discovery of borneol dates back to ancient times, where it was used in Chinese medicine as a remedy for various ailments. It was initially derived from the camphor tree (*Cinnamomum camphora*) but has since been found in other plant species. Its isolation and structure were well-established in the 19th century, following advancements in organic chemistry. (+)-Borneol has a wide range of applications, particularly in the fragrance and pharmaceutical industries. As a key component of essential oils, it contributes to the pleasant odor profile of many herbal and floral products. Its distinct camphor-like aroma has made it a valuable ingredient in perfumes, cosmetics, and personal care products. In traditional medicine, borneol has been used for its purported therapeutic properties, including its ability to enhance circulation, reduce inflammation, and alleviate pain. In modern pharmacology, it is recognized for its potential as a carrier or enhancer in drug delivery systems. Its ability to enhance the permeability of biological membranes has been explored in the context of improving the bioavailability of various drugs. (+)-Borneol has also been studied for its antimicrobial properties. Research has shown that it exhibits activity against a range of microorganisms, including bacteria and fungi. This has led to its incorporation into topical formulations for treating infections and promoting wound healing. Additionally, borneol has been investigated for its potential to act as an anti-inflammatory and analgesic agent in various clinical contexts. In addition to its use in medicine, (+)-borneol is employed in the synthesis of other valuable chemical compounds, including camphor and menthol, through chemical transformations. The synthesis of borneol itself can be achieved through several methods, including the reduction of camphor or by direct biosynthesis from natural precursors. Overall, (+)-borneol is a well-established compound in both traditional and modern applications, recognized for its therapeutic properties, fragrance contributions, and utility in the pharmaceutical and cosmetic industries. Its continued use is supported by extensive research and historical precedent. References 2024. Nasal administration of Xingnaojing biomimetic nanoparticles for the treatment of ischemic stroke. International Journal of Pharmaceutics, 667. DOI: 10.1016/j.ijpharm.2024.124830 2024. Experiments Validated the Development of Zebrafish Embryos and Toxicological Mechanism of Borneols in Perinatal Period. Current Pharmaceutical Design, 30(41). DOI: 10.2174/0113816128319753240730052138 2024. Metabolic engineering of the borneol and camphor degradation pathways in Pseudomonas to produce optically pure bicyclic monoterpenes. Enzyme and Microbial Technology, 181. DOI: 10.1016/j.enzmictec.2024.110527 |
| Market Analysis Reports |