Online Database of Chemicals from Around the World
| Changzhou Share Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sharechem.com | |||
 | +86 (519) 8261-9998 | |||
 | +86 (519) 8261-9997 | |||
 | wu@sharechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Zhangjiagang Pharmabl Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharmabl.com | |||
 | +86 (512) 5818-9390 | |||
 | +86 (512) 5818-1490 | |||
 | info@pharmabl.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Thirumalai Chemicals Ltd | India | |||
|---|---|---|---|---|
 | www.thirumalaichemicals.com | |||
 | +91 (41) 7224-4441 | |||
 | +91 (41) 7224-4308 | |||
 | info@thirumalaichemicals.com | |||
| Chemical manufacturer since 1976 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Fenhe Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fenhechem.com | |||
 | +86 (021) 3392-6068 | |||
 | julius.wei@fenhechem.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Organo Fine Chemicals | India | |||
|---|---|---|---|---|
 | www.organo.in | |||
 | +91 (22) 2201-6968 2206-7521 | |||
 | +91 (22) 2205-9299 / 2208-3184 | |||
 | info@organo.in | |||
| Chemical manufacturer | ||||
| Zhangjiagang Xinyi Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xinyichem.com | |||
 | +86 (512) 5866-7398 5859-1449 | |||
 | +86 (512) 5859-1449 | |||
 | zjgxyhg@126.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Fatty acid |
|---|---|
| Name | Propiolic acid |
| Synonyms | 2-Propynoic acid; Acetylenecarboxylic acid; Propargylic acid |
| Molecular Structure | 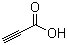 |
| Molecular Formula | C3H2O2 |
| Molecular Weight | 70.05 |
| CAS Registry Number | 471-25-0 |
| EC Number | 207-437-8 |
| SMILES | C#CC(=O)O |
| Density | 1.138 |
|---|---|
| Melting point | 16 °C |
| Boiling point | 102 °C (200 mmHg) |
| Refractive index | 1.4315-1.4335 |
| Flash point | 58 °C |
| Water solubility | miscible |
| Hazard Symbols |    GHS02;GHS05;GHS06 Danger Details GHS02;GHS05;GHS06 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H302-H310-H314 Details | ||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P260-P262-P264-P270-P280-P301+P316-P301+P330+P331-P302+P352-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P321-P330-P361+P364-P363-P370+P378-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| Transport Information | UN 2920 | ||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Propiolic acid, also known as 2-propynoic acid, is a simple organic compound with the formula HC≡C-COOH. This compound is notable for its triple bond, making it an alkyne carboxylic acid. It has garnered significant interest in organic chemistry due to its unique reactivity and versatile applications in various fields, including pharmaceuticals, materials science, and chemical synthesis. The discovery of propiolic acid dates back to the 19th century during the exploration of alkynes and carboxylic acids. Chemists identified propiolic acid as a compound that could be synthesized through the oxidation of propargyl alcohol or by the hydration of acetylene under specific conditions. The presence of both a carboxyl group and a triple bond in its structure made it a subject of interest for chemists looking to explore its potential reactivity and applications. One of the most prominent features of propiolic acid is its alkyne triple bond, which imparts significant reactivity to the molecule. This triple bond allows propiolic acid to undergo a variety of chemical reactions, making it a valuable building block in organic synthesis. For example, the triple bond can participate in addition reactions, where it reacts with various reagents to form new carbon-carbon or carbon-heteroatom bonds. This reactivity is particularly useful in the synthesis of more complex molecules, such as pharmaceuticals, agrochemicals, and specialty chemicals. In the pharmaceutical industry, propiolic acid is employed as an intermediate in the synthesis of various drugs. Its ability to form new bonds through its triple bond allows for the construction of molecules with intricate structures, which are often required in drug design. Additionally, derivatives of propiolic acid have been investigated for their biological activities, including potential anticancer and antimicrobial properties. The triple bond in these derivatives is thought to play a key role in their interactions with biological targets, making propiolic acid an important precursor in medicinal chemistry. Propiolic acid also has applications in materials science, particularly in the development of polymers and advanced materials. The reactivity of the alkyne group allows propiolic acid to be used in polymerization reactions, leading to the formation of materials with unique properties. These materials can be tailored for specific applications, such as coatings, adhesives, and electronic devices. The ability to introduce a carboxylic acid group into these materials further enhances their functionality, making propiolic acid a valuable component in the creation of innovative materials. Moreover, propiolic acid is utilized in organic synthesis as a versatile reagent. It can be used to introduce the alkyne functionality into various molecules, enabling the synthesis of a wide range of compounds with different chemical properties. This versatility makes it a key reagent in the toolbox of synthetic chemists, who use it to build complex molecules for research and industrial purposes. In summary, propiolic acid is a chemically significant compound with wide-ranging applications in organic synthesis, pharmaceuticals, and materials science. Its discovery and subsequent utilization have provided chemists with a valuable tool for constructing complex molecules and developing advanced materials. The unique reactivity of its triple bond continues to make propiolic acid a compound of interest in both scientific research and industrial applications. References 2023. Copper-catalyzed C-C bond formation via carboxylation reactions with CO2. C-C Cross Couplings with 3d Base Metal Catalysts, 82. DOI: 10.1007/3418_2023_82 2024. Decarboxylative click cycloaddition: An emerging strategy towards substituted 1,2,3-triazole derivatives. Molecular Diversity, 28. DOI: 10.1007/s11030-024-11014-4 1959. Condensation of 1-vinylcyclohexene with propiolic and tetrolic acids and esters. Bulletin of the Academy of Sciences of the USSR, Division of Chemical Science, 8. DOI: 10.1007/bf00914221 |
| Market Analysis Reports |