Online Database of Chemicals from Around the World
| Yancheng Chemhome Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemhome.com.cn | |||
 | +86 (515) 8330-1301 | |||
 | +86 (515) 8820-3699 | |||
 | home@chemhome.com.cn | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2006 | ||||
| Yizheng Haifan Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yzhaifan.com | |||
 | +86 (514) 8346-8749 | |||
 | +86 (514) 8266-0277 | |||
 | zcw@yzhaifan.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Papchem Lifesciences | India | |||
|---|---|---|---|---|
 | papchemlifesciences.com | |||
 | 9061328401 | |||
 | bd@papchemlifesciences.com | |||
| Chemical manufacturer since 2021 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Chemical pesticide >> Insecticide intermediate |
|---|---|
| Name | 3,4,5,6-Tetrahydrophthalimide |
| Synonyms | 1-Cyclohexene-1,2-dicarboximide |
| Molecular Structure | 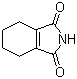 |
| Molecular Formula | C8H9NO2 |
| Molecular Weight | 151.16 |
| CAS Registry Number | 4720-86-9 |
| EC Number | 225-211-7 |
| SMILES | C1CCC2=C(C1)C(=O)NC2=O |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H319 Details | ||||||||||||
| Safety Statements | P264-P264+P265-P270-P280-P301+P317-P305+P351+P338-P330-P337+P317-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| Transport Information | UN 2811 | ||||||||||||
| SDS | Available | ||||||||||||
|
3,4,5,6-Tetrahydrophthalimide* is an important compound with notable applications in organic synthesis and pharmaceuticals. Its unique structure and properties make it a versatile building block in various chemical processes. 3,4,5,6-Tetrahydrophthalimide was first synthesized in the mid-20th century as part of the study of polycyclic compounds and their derivatives. The compound is derived from phthalimide, where the aromatic ring is partially hydrogenated, resulting in a tetrahydro derivative. The synthesis involves the reduction of the aromatic ring, usually using catalytic hydrogenation methods. 3,4,5,6-Tetrahydrophthalimide can be used as an intermediate in the synthesis of various drugs. Its unique structure allows the formation of complex molecules with potential therapeutic effects. It is particularly useful in the development of compounds with specific biological activities. In organic chemistry, 3,4,5,6-Tetrahydrophthalimide is a versatile reagent used to build various organic frameworks. It is used in reactions such as cyclization and functional group transformations, helping to produce complex molecules. The compound is used in the development of agrochemicals, including herbicides and pesticides. Its properties can be used to improve the efficacy and stability of these products, thereby increasing agricultural productivity. 3,4,5,6-Tetrahydrophthalimide is involved in the synthesis of dyes and pigments. Its role as a building block in these applications helps produce vibrant and stable colors used in various industries. In materials science, the compound is explored for its potential use in the development of polymers and other materials. Its chemical properties help form materials with desirable properties, such as enhanced stability or flexibility. The unique tetrahydro structure of 3,4,5,6-Tetrahydrophthalimide lends itself to versatility in organic synthesis. Its ability to participate in a wide range of chemical reactions makes it a valuable tool for the creation of a wide range of compounds. Its role as an intermediate in drug synthesis highlights its importance in drug development. The structural features of the compound make it possible to synthesize bioactive molecules with potential therapeutic benefits. 3,4,5,6-Tetrahydrophthalimide exhibits good stability and reactivity, making it suitable for use in a variety of chemical processes. Its stability ensures its reliable performance in both industrial and laboratory settings. References 2012. Toxicokinetic modeling of captan fungicide and its tetrahydrophthalimide biomarker of exposure in humans. Toxicology Letters, 208. DOI: 10.1016/j.toxlet.2011.09.023 2007. Antinociceptive effects of tetrahydrophthalimides and related compounds. Zeitschrift fur Naturforschung C, 62. DOI: 10.1515/znc-2007-3-408 1993. Captan metabolism in humans yields two biomarkers, tetrahydrophthalimide (THPI) and thiazolidine-2-thione-4-carboxylic acid (TTCA) in urine. Drug and Chemical Toxicology, 16. DOI: 10.3109/01480549309031997 |
| Market Analysis Reports |