Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Okeanos Tech. Jiangsu Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.okeanospharm.com | |||
 | +86 (510) 8663-6249 | |||
 | +86 (510) 8609-3680 | |||
 | sales@okeanospharm.com | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Allbiosyn Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.allbiosyn.com | |||
 | +86 15900695956 | |||
 | info@allbiosyn.com | |||
 | WeChat: 15900695956 | |||
 | WhatsApp:86 15900695956 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2024 | ||||
| W & J PharmaChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.wjpharmachem.com | |||
 | +1 (301) 880-0624 | |||
 | +1 (301) 434-9366 | |||
 | info@wjpharmachem.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Nucleoside drugs >> Nucleoside intermediate |
|---|---|
| Name | 2'-O-(2-Methoxyethyl)guanosine |
| Synonyms | 2-amino-9-[(2R,3R,4R,5R)-4-hydroxy-5-(hydroxymethyl)-3-(2-methoxyethoxy)oxolan-2-yl]-1H-purin-6-one |
| Molecular Structure | 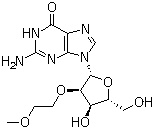 |
| Molecular Formula | C13H19N5O6 |
| Molecular Weight | 341.32 |
| Protein Sequence | N |
| CAS Registry Number | 473278-54-5 |
| SMILES | COCCO[C@@H]1[C@@H]([C@H](O[C@H]1N2C=NC3=C2N=C(NC3=O)N)CO)O |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302 Details |
| Safety Statements | P280-P305+P351+P338 Details |
| SDS | Available |
|
2'-O-(2-methoxyethyl)guanosine is a well-known nucleoside derivative with important significance in nucleic acid chemistry and therapeutic development. This compound, also known as 2'-O-(2-methoxyethyl)guanosine, is valued for its stability and versatility in various chemical and biological applications. The synthesis of 2'-O-(2-methoxyethyl)guanosine stems from the need to develop nucleoside analogs with enhanced stability and biological activity. Researchers aimed to modify the ribose component of guanosine to improve its resistance to enzymatic degradation and increase its solubility in aqueous environments. The introduction of a 2-methoxyethyl group at the 2' position of the ribose ring proved to be effective in achieving these goals. The compound was first reported in the early 2000s, and its synthesis involves the selective protection of the guanine base followed by the introduction of the 2-methoxyethyl group. This modification provides the nucleoside with a stable yet functional structure suitable for further chemical manipulations. 2'-O-(2-methoxyethyl)guanosine is widely used in the synthesis of modified oligonucleotides. The methoxyethyl group enhances the stability of nucleosides against nucleases, making them ideal for therapeutic oligonucleotides and diagnostic probes. This increased stability prolongs the half-life of oligonucleotides in biological systems, improving their efficacy and reliability in a variety of applications. In antiviral and anticancer research, 2'-O-(2-methoxyethyl)guanosine is a key component in the development of nucleoside analogs with potential therapeutic properties. By modifying the guanosine structure, researchers can create compounds that exhibit enhanced potency and selectivity against viral enzymes or cancer cells. These analogs may provide new avenues for the development of drugs with improved pharmacological profiles. The compound is also used as a building block for the synthesis of specialized RNA sequences in molecular biology. Its stability makes it valuable for creating RNA molecules that resist degradation, aiding the study of RNA function and interactions. In addition, it is used in the development of RNA-based therapeutics, including ribozymes and RNA interference technologies. References 2002. Synthesis of 2‘-O-Methoxyethylguanosine Using a Novel Silicon-Based Protecting Group. The Journal of Organic Chemistry, 67(20). DOI: 10.1021/jo025970e 2008. Process Research on the Preparation of DMT Protected 2′-O-Methoxyethylguanosine for Oligonucleotide Synthesis in Therapeutic Applications. Nucleosides, Nucleotides and Nucleic Acids, 27(9). DOI: 10.1080/15257770802271748 |
| Market Analysis Reports |