Online Database of Chemicals from Around the World
| Changzhou Jiuwu Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiuwuchem.com | |||
 | +86 (519) 8512-1086 +86 13775162768 | |||
 | +86 (519) 8512-5369 | |||
 | info@jiuwuchem.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2016 | ||||
| Classification | Chemical pesticide >> Insecticide intermediate |
|---|---|
| Name | 4-Ethoxyphenylacetic acid |
| Synonyms | 2-(4-ethoxyphenyl)acetic acid |
| Molecular Structure | 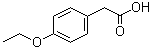 |
| Molecular Formula | C10H12O3 |
| Molecular Weight | 180.20 |
| CAS Registry Number | 4919-33-9 |
| EC Number | 225-545-3 |
| SMILES | CCOC1=CC=C(C=C1)CC(=O)O |
| Solubility | Slightly soluble (2.7 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.144±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 91.3 °C** |
| Boiling point | 318.5±17.0 °C (760 Torr), Calc.* |
| Flash point | 125.8±14.4 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2013 ACD/Labs) |
| ** | Profft, E. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
4-Ethoxyphenylacetic acid (C9H12O3) is an organic compound that consists of a phenylacetic acid backbone with an ethoxy group (-OCH2CH3) attached to the para-position (4-position) of the aromatic ring. It is a derivative of phenylacetic acid, a compound that has found applications in various fields, particularly in the synthesis of pharmaceuticals, agrochemicals, and specialty chemicals. The compound is typically synthesized through esterification reactions, where 4-ethoxyphenol is reacted with acetic acid or its derivatives, followed by hydrolysis to yield the desired acid. The discovery of 4-ethoxyphenylacetic acid is part of an ongoing effort to explore modified phenylacetic acids, which are versatile intermediates in organic chemistry. The ethoxy group in this compound not only influences its chemical properties but also enhances its solubility and stability, making it more suitable for different chemical applications. The compound is often synthesized as part of a larger series of substituted phenylacetic acids, which are of interest due to their potential biological and chemical activity. One of the most significant applications of 4-ethoxyphenylacetic acid is in the pharmaceutical industry, where it serves as a key intermediate in the synthesis of various drugs. The compound's structure is highly suitable for modifications that can lead to compounds with anti-inflammatory, analgesic, or antimicrobial properties. 4-Ethoxyphenylacetic acid and its derivatives are often used to design prodrugs or act as a scaffold for drug development. For example, the compound can be modified to create inhibitors or modulators of enzymes involved in pain and inflammation pathways. Beyond pharmaceuticals, 4-ethoxyphenylacetic acid has also found use in the agrochemical industry. It serves as a building block for the synthesis of herbicides and insecticides. The compound can be easily functionalized to introduce different groups that enhance its biological activity. In particular, 4-ethoxyphenylacetic acid derivatives are employed in the design of selective herbicides that target specific enzymes in weeds, providing a safer and more efficient alternative to traditional herbicides. In addition to its uses in pharmaceuticals and agrochemicals, 4-ethoxyphenylacetic acid has been studied for its role in the production of specialty chemicals, including fragrances and polymer additives. Its aromatic structure allows it to serve as a precursor for more complex molecules, including those with potential use in materials science, where its ethoxy group can enhance the properties of the final products. The compound is also an important reagent in organic synthesis, where it is involved in reactions such as alkylation and acylation, which are central to the creation of diverse organic compounds. In summary, 4-ethoxyphenylacetic acid is an important compound with applications in several industries, including pharmaceuticals, agrochemicals, and materials science. Its versatility as a building block for the synthesis of bioactive molecules and functional materials makes it a valuable intermediate in organic chemistry. The ongoing development of 4-ethoxyphenylacetic acid and its derivatives continues to offer promising opportunities in drug discovery, pest control, and other chemical applications. References 2024. Fragment library screening by X-ray crystallography and binding site analysis on thioredoxin glutathione reductase of Schistosoma mansoni. Scientific Reports. DOI: PMC10796382 2022. Structural Analysis and Development of Notum Fragment Screening Hits. ACS Chemical Neuroscience, 13(13). DOI: PMC9264368 2008. 1-(5-Chloro-2,4-dihydroxy-phen-yl)-2-(4-ethoxy-phen-yl)ethanone. Acta crystallographica. Section E, Structure reports online. DOI: PMC2959265 |
| Market Analysis Reports |