Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Shanghai Oripharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.oripharm.com.cn | |||
 | +86 (21) 6439-6936 | |||
 | +86 (21) 6481-4386 | |||
 | info@oripharm.com.cn sales@oripharm.com.cn | |||
| Chemical distributor since 2003 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Beijing Ribio Biotech. Inc. | China | |||
|---|---|---|---|---|
 | www.ribio.com.cn | |||
 | +86 (10) 6266-4360 +86 13910960639 | |||
 | +86 (10) 6266-4370 | |||
 | sales@ribio.com.cn ribio@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Quzhou HuntBio Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huntchem.com | |||
 | +86 (571) 6406-7827 +86 13868119375 | |||
 | +86 (571) 6479-8288 | |||
 | sales@huntchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Selleck Chemicals LLC | USA | |||
|---|---|---|---|---|
 | www.selleckchem.com | |||
 | +1 (713) 535-9129 | |||
 | +1 (832) 582-8590 | |||
 | info@selleckchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Changzhou Litong Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.litonchem.com | |||
 | +86 (519) 8630-1238 | |||
 | +86 (519) 8659-1192 | |||
 | htszh2004@163.com | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Tecoland Corporation | USA | |||
|---|---|---|---|---|
 | www.tecoland.com | |||
 | +1 (732) 603-9577 | |||
 | +1 (732) 906-1522 | |||
 | info@tecoland.com | |||
| Chemical distributor since 2001 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Aajiang Jiuzhou Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiuzhou-chem.com | |||
 | +86 13454675544 | |||
 | jamie@jiuzhou-chem.com | |||
 | QQ Chat | |||
 | WeChat: +86 18632988332 | |||
 | WhatsApp:+86 18632988332 | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Melford Laboratories Limited | UK | |||
|---|---|---|---|---|
 | www.melford.co.uk | |||
 | +44 (1449) 741-178 | |||
 | +44 (1449) 741-217 | |||
 | pfranklin@melford.co.uk sales@melford.co.uk | |||
| Chemical manufacturer since 1985 | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Berry & Associates, Inc. | USA | |||
|---|---|---|---|---|
 | www.berryassoc.com | |||
 | +1 (734) 426-3787 | |||
 | +1 (734) 426-9077 | |||
 | orders@berryassoc.com | |||
| Chemical manufacturer | ||||
| AK Scientific, Inc | USA | |||
|---|---|---|---|---|
 | www.aksci.com | |||
 | +1 (510) 429-8835 | |||
 | +1 (510) 429-8836 | |||
 | sales@aksci.com | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Antineoplastic agents >> Antimetabolite antineoplastic |
|---|---|
| Name | Floxuridine |
| Synonyms | Fluorodeoxyuridine; Fluoruridine deoxyribose; 2'-Deoxy-5-fluorouridine; (+)-5-Fluoro-2'-deoxyuridine |
| Molecular Structure | 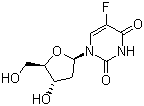 |
| Molecular Formula | C9H11FN2O5 |
| Molecular Weight | 246.19 |
| Protein Sequence | N |
| CAS Registry Number | 50-91-9 |
| EC Number | 200-072-5 |
| SMILES | C1[C@@H]([C@H](O[C@H]1N2C=C(C(=O)NC2=O)F)CO)O |
| Solubility | Soluble 100 mM (water), 100 mM (DMSO) (Expl.) |
|---|---|
| Density | 1.6±0.1 g/cm3, Calc.* |
| Melting point | 148 °C (Expl.) |
| alpha | 35.9 ° (c=1, water) |
| Index of Refraction | 1.609, Calc.* |
| Boiling Point | 150 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS06;GHS08 Danger Details GHS06;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H341 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P264-P270-P280-P301+P316-P318-P321-P330-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2811 | ||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
Floxuridine is a chemotherapeutic agent used primarily in the treatment of cancer. It is a synthetic nucleoside analog of thymidine and belongs to the class of drugs known as antimetabolites. Floxuridine is utilized in the treatment of colorectal cancer and other solid tumors, particularly when the cancer has metastasized to the liver. Floxuridine was first synthesized and introduced in the mid-20th century as part of the efforts to develop drugs that could interfere with DNA synthesis and inhibit cancer cell proliferation. Its structure, a fluorinated derivative of deoxyuridine, was designed to mimic the nucleosides naturally involved in DNA replication. The presence of the fluorine atom in the molecule gives it its therapeutic properties, as it can be incorporated into the DNA of rapidly dividing cells, leading to DNA damage and cell death. The action of floxuridine is primarily through its interference with thymidylate synthase, an enzyme critical for the synthesis of thymidine monophosphate, a precursor to thymidine, one of the four building blocks of DNA. When floxuridine is incorporated into the DNA of cancer cells, it disrupts DNA replication and leads to cell death, specifically targeting cells that are rapidly dividing, such as tumor cells. This mechanism of action makes floxuridine effective in the treatment of cancers that are characterized by uncontrolled cell proliferation. Floxuridine is typically administered as an intravenous injection or infused into the hepatic artery, especially for cancers that have metastasized to the liver. This method of administration ensures that the drug is delivered directly to the liver, maximizing its effectiveness while minimizing systemic side effects. The drug is often used in conjunction with other cancer treatments, such as surgery, radiation therapy, and other chemotherapeutic agents, to improve overall treatment outcomes. The drug is commonly used in the treatment of liver metastases from colorectal cancer. It has been shown to be effective in reducing the size of tumors and alleviating symptoms in patients with advanced liver metastases. Floxuridine has also been studied for its potential in treating other solid tumors, including those of the pancreas and stomach. In some cases, it is used as part of a palliative treatment regimen to improve quality of life for patients with advanced cancer. Floxuridine, like other chemotherapy agents, can have significant side effects. These include bone marrow suppression, nausea, vomiting, and gastrointestinal distress. The drug’s effects on rapidly dividing cells are not limited to cancer cells, which can lead to damage to normal, healthy cells, particularly those in the gastrointestinal tract and bone marrow. As a result, its use requires careful monitoring, particularly for signs of myelosuppression, a reduction in the production of blood cells. Despite these potential side effects, floxuridine has been an important tool in the management of certain cancers, particularly when liver metastasis is involved. In conclusion, floxuridine is a vital chemotherapeutic agent used in the treatment of cancers, especially those with liver metastases. Its mechanism of action involves the disruption of DNA synthesis, leading to the death of rapidly dividing cancer cells. While it is an effective treatment for specific types of cancer, its use requires careful monitoring due to potential side effects. The development of floxuridine marked an important advance in cancer therapy, providing a valuable option for patients with advanced disease. References 1994. Phase I and pharmacokinetic evaluation of floxuridine/leucovorin given on the Roswell Park weekly regimen. Cancer Chemotherapy and Pharmacology, 34(4). DOI: 10.1007/bf00685087 1998. In vitro study on intrathecal use of 5-fluoro-2′-deoxyuridine (FdUrd) for meningeal dissemination of malignant brain tumors. Journal of Neuro-Oncology, 37(3). DOI: 10.1023/a:1005869226496 2000. Raman Spectroscopy of Uracil DNA Glycosylase−DNA Complexes: Insights into DNA Damage Recognition and Catalysis. Biochemistry, 39(40). DOI: 10.1021/bi001437m |
| Market Analysis Reports |