Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Watec Laboratories, Inc. | China | |||
|---|---|---|---|---|
 | www.wateclaboratories.com | |||
 | +86 (519) 8692-1516 +86 18602586511 | |||
 | +86 (519) 8526-7382 | |||
 | info@wateclaboratories.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| R&D Scientific Inc. | Canada | |||
|---|---|---|---|---|
 | www.rdscientific.com | |||
 | +1 (226) 600-0236 | |||
 | sales@rdscientific.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| GLSynthesis Inc. | USA | |||
|---|---|---|---|---|
 | www.glsynthesis.com | |||
 | +1 (508) 754-6700 | |||
 | +1 (508) 754-7075 | |||
 | support@glsynthesis.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Amine |
|---|---|
| Name | 4-Vinylbenzylamine |
| Synonyms | (4-ethenylphenyl)methanamine |
| Molecular Structure | 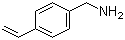 |
| Molecular Formula | C9H11N |
| Molecular Weight | 133.19 |
| CAS Registry Number | 50325-49-0 |
| EC Number | 678-341-5 |
| SMILES | C=CC1=CC=C(C=C1)CN |
| Density | 1.0$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 231.4$+/-$9.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 96.0$+/-$8.5 $degree$C (Calc.)* |
| Index of refraction | 1.589 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
4-Vinylbenzylamine is an aromatic amine containing both a primary amine functional group and a vinyl-substituted benzyl moiety. Its molecular formula is C9H11N. The structure consists of a benzene ring bearing a benzylic methylene amine group (–CH2NH2) at one position and a vinyl group (–CH=CH2) at the para (4-) position. The molecule combines three key structural features: an aromatic ring, a primary amine, and a terminal alkene. The benzene ring provides aromatic stability and serves as a rigid scaffold. The benzylic amine group introduces basicity and nucleophilicity, while the vinyl substituent provides an unsaturated site capable of undergoing addition or polymerization reactions. The amine functionality is a primary aliphatic amine attached to a benzylic carbon, which increases its reactivity compared with aniline-type aromatic amines. It can readily undergo protonation to form ammonium salts, acylation to form amides, alkylation, and condensation reactions with carbonyl compounds to form imines or related derivatives. The vinyl group at the para position is conjugated through the aromatic ring only weakly, but it remains an important reactive handle. It can participate in radical polymerization, allowing the compound to act as a monomer or comonomer in polymer synthesis. The presence of both an amine and a vinyl group makes 4-vinylbenzylamine a bifunctional building block for functional polymer materials. From a structural perspective, the benzylamine portion (–CH2NH2) is flexible and can freely rotate relative to the aromatic ring. The para-vinyl substituent introduces additional reactivity without significantly hindering the amine functionality. This combination allows selective chemical modification at either functional group depending on reaction conditions. 4-Vinylbenzylamine is typically synthesized through multi-step organic reactions starting from substituted benzyl derivatives. One common approach involves preparation of 4-vinylbenzyl halides followed by nucleophilic substitution with ammonia or amine sources to introduce the benzylamine functionality. Alternatively, reduction of corresponding nitrile or imine precursors can also yield the amine. In polymer chemistry, 4-vinylbenzylamine is used as a functional monomer or comonomer to introduce reactive amine groups into polymer backbones. The vinyl group participates in free-radical polymerization, while the pendant amine group remains available for post-polymerization modification, such as crosslinking, grafting, or ion exchange reactions. The presence of primary amine functionality makes polymers derived from this monomer useful in applications such as ion-exchange resins, surface modification, and biomolecule immobilization. The amine groups can interact with acids, metal ions, or biological molecules through electrostatic interactions or covalent coupling chemistry. From a physicochemical perspective, 4-vinylbenzylamine exhibits moderate polarity due to the amine group, while the aromatic ring and vinyl substituent contribute hydrophobic character. The compound is capable of hydrogen bonding through the amine hydrogen atoms, influencing its solubility and intermolecular interactions. The vinyl group makes the molecule sensitive to radical initiation and polymerization conditions, particularly in the presence of peroxides or other radical initiators. Careful control is required to prevent undesired polymerization during storage or handling. Historically, vinyl-substituted benzylamines have been important in the development of functional polymers with tailored chemical reactivity. Their dual functionality enables incorporation into polymer backbones while preserving chemically active side groups. Overall, 4-vinylbenzylamine is a bifunctional aromatic compound containing a primary benzylamine group and a para-vinyl substituent. Its combination of polymerizable unsaturation and reactive amine functionality makes it a valuable monomer in polymer chemistry, surface functionalization, and materials science applications. References 2019. Facile Synthesis of Boronate Affinity-Based Molecularly Imprinted Monolith with Reduced Capturing pH Towards Cis-Diol-Containing Compounds. Chromatographia. DOI: 10.1007/s10337-019-03736-7 |
| Market Analysis Reports |