Online Database of Chemicals from Around the World
| Shanghai Tajilin Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tajilin.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales003@tajilin.com | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Ryan Scientific, Inc. | USA | |||
|---|---|---|---|---|
 | www.ryansci.com | |||
 | +1 (843)-884-4911 | |||
 | +1 (843) 884-5568 | |||
 | sales@ryansci.com | |||
| Chemical manufacturer | ||||
| Advanced Chemical Intermediates Ltd. | UK | |||
|---|---|---|---|---|
 | www.acints.com | |||
 | +44 (1840) 261-451 | |||
 | +44 (1840) 261-451 | |||
 | enquiries@acints.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Hydrocarbon compounds and their derivatives >> Hydrocarbon halide |
|---|---|
| Name | 2-Chloro-1,1,1-triethoxyethane |
| Synonyms | Triethyl 2-chloroorthoacetate |
| Molecular Structure | 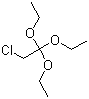 |
| Molecular Formula | C8H17ClO3 |
| Molecular Weight | 196.67 |
| CAS Registry Number | 51076-95-0 |
| EC Number | 628-654-8 |
| SMILES | CCOC(CCl)(OCC)OCC |
| Density | 1.031 |
|---|---|
| Boiling point | 75-80 °C (15 mmHg) |
| Refractive index | 1.4215-1.4235 |
| Flash point | 27 °F |
| Hazard Symbols |   GHS02;GHS07 Danger Details GHS02;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H302+H315+H317-H319-H335 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P261-P264-P264+P265-P270-P271-P272-P280-P301+P317-P302+P352-P303+P361+P353-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P333+P317-P337+P317-P362+P364-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1993 | ||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
2-Chloro-1,1,1-triethoxyethane, also known as ethyl chloro(hydroxy)acetate, was first synthesized and identified in the field of organic chemistry in the mid-20th century. Its discovery was a result of research into the synthesis and properties of chlorinated organic compounds. This chemical compound is characterized by a chloro group attached to a carbon atom, along with three ethoxy groups bonded to the same carbon atom. Its synthesis and structure were elucidated through organic synthesis techniques and spectroscopic analysis, contributing to the understanding of organic chemistry principles. 2-Chloro-1,1,1-triethoxyethane serves as a valuable intermediate in organic synthesis. It participates in various chemical reactions, including nucleophilic substitution and esterification reactions, to form complex organic molecules. It is commonly used in the production of pharmaceuticals, agrochemicals, and fine chemicals due to its versatile reactivity and ability to introduce functional groups into organic compounds. This compound finds applications as a solvent in various industrial processes. It serves as a reaction medium for organic transformations, such as alkylations and acylations, in chemical synthesis. Its use as a solvent enables the dissolution and purification of organic compounds in laboratory and industrial settings. Additionally, it acts as a versatile chemical reagent for the modification of organic molecules in diverse applications. 2-Chloro-1,1,1-triethoxyethane is utilized in esterification reactions to introduce chloroalkyl groups into organic compounds. This chemical transformation is valuable in the synthesis of specialty chemicals and pharmaceutical intermediates. The presence of the chloro group enables further derivatization of the resulting ester, expanding its utility in various applications. This compound is employed in the formulation of surface coatings, such as paints, varnishes, and adhesives. It contributes to the chemical composition of coatings, providing desired properties such as adhesion, durability, and corrosion resistance. 2-Chloro-1,1,1-triethoxyethane may be used as a solvent or coalescing agent in coating formulations, enhancing film formation and improving coating performance. In the polymer industry, 2-Chloro-1,1,1-triethoxyethane is utilized in the synthesis of specialty polymers and copolymers. It acts as a monomer or comonomer in polymerization reactions, contributing to the formation of polymers with specific properties and functionalities. Its incorporation into polymer chains enhances the mechanical, thermal, and chemical properties of the resulting materials, making them suitable for various applications, including coatings, adhesives, and sealants. References 2017. Synthesis of tetrazole and its derivatives by heterocyclization reaction involving primary amines, orthoesters, and azides. Chemistry of Heterocyclic Compounds, 53(6-7). DOI: 10.1007/s10593-017-2108-7 2005. Synthesis of the EF Fragment of Spongistatin 1. Synlett, 2005(12). DOI: 10.1055/s-2005-871960 |
| Market Analysis Reports |