Online Database of Chemicals from Around the World
| Shanghai Rui Yun Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rychemical.com.cn | |||
 | +86 (21) 6726-7633 | |||
 | +86 (21) 6726-7655 | |||
 | sales@rychemical.com.cn | |||
 | WeChat: 13917251563 | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2009 | ||||
| Classification | Chemical reagent >> Organic reagent >> Ether |
|---|---|
| Name | Ethyl 1,1,2,2-tetrafluoroethyl ether |
| Synonyms | 1,1,2,2-Tetrafluoroethyl ethyl ether |
| Molecular Structure | 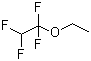 |
| Molecular Formula | C4H6F4O |
| Molecular Weight | 146.08 |
| CAS Registry Number | 512-51-6 |
| EC Number | 680-776-0 |
| SMILES | CCOC(C(F)F)(F)F |
| Density | 1.198 g/mL |
|---|---|
| Boiling point | 56-58 °C |
| Flash point | -15 °C |
| Hazard Symbols |   GHS02;GHS07 Danger Details GHS02;GHS07 Danger Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P261-P264-P264+P265-P271-P280-P302+P352-P303+P361+P353-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Ethyl 1,1,2,2-tetrafluoroethyl ether, with the chemical formula C₄H₅F₄O, is a specialized fluorinated compound notable for its unique chemical properties and diverse applications. The synthesis of ethyl 1,1,2,2-tetrafluoroethyl ether dates back to the mid-20th century, as researchers sought to explore and develop fluorinated ethers for their distinct physical and chemical characteristics. Its structure features an ethyl group attached to a tetrafluoroethyl moiety, making it a highly fluorinated organic compound. In the chemical industry, ethyl 1,1,2,2-tetrafluoroethyl ether is used primarily as a solvent due to its high chemical stability and low reactivity. These properties make it suitable for use in various industrial processes where a non-reactive solvent is required. It is particularly useful in the production and formulation of specialty chemicals and pharmaceuticals. The compound also finds applications in the field of material science. Its unique fluorinated structure imparts certain desirable properties, such as low surface tension and high chemical resistance, which are valuable in the development of advanced materials. For instance, it is used in the manufacture of coatings and polymers that require enhanced durability and resistance to chemical attack. Additionally, ethyl 1,1,2,2-tetrafluoroethyl ether is utilized in the synthesis of other fluorinated compounds. Its ability to act as a reagent in organic synthesis allows chemists to create new molecules with tailored properties for specific applications. This versatility makes it a valuable tool in the development of innovative materials and chemicals. Research into the environmental and health impacts of fluorinated compounds, including ethyl 1,1,2,2-tetrafluoroethyl ether, is ongoing. Its persistence in the environment and potential effects on health are subjects of interest, particularly given the broader context of fluorinated chemicals' environmental impact. References 2024. Recent advances in li metal anode protection for high performance lithium-sulfur batteries. Discover Chemical Engineering, 4(1). DOI: 10.1007/s43938-024-00045-w 2021. LiFSI as a functional additive of the fluorinated electrolyte for rechargeable Li-S batteries. Journal of Materials Science: Materials in Electronics, 32(4). DOI: 10.1007/s10854-021-05310-0 2018. Recent Progress in Liquid Electrolyte-Based Li-S Batteries: Shuttle Problem and Solutions. Electrochemical Energy Reviews, 1(4). DOI: 10.1007/s41918-018-0021-0 |
| Market Analysis Reports |