Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Zhejiang Qiming Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qimingpharm.com | |||
 | +86 (571) 8716-3895 8716-3893 | |||
 | +86 (571) 8716-3816 | |||
 | sales@qimingpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Xinxiang Aurora Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aurora-biotech.com | |||
 | +86 (373) 308-8722 +86 18637352520 | |||
 | +86 (373) 308-8722 | |||
 | info@aurora-biotech.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Chemical reagent >> Organic reagent >> Ester >> Ethyl ester compound |
|---|---|
| Name | Ethyl 2,3,4,5-tetrafluorobenzoyl acetate |
| Synonyms | ethyl 3-oxo-3-(2,3,4,5-tetrafluorophenyl)propanoate |
| Molecular Structure | 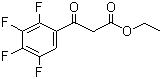 |
| Molecular Formula | C11H8F4O3 |
| Molecular Weight | 264.17 |
| CAS Registry Number | 94695-50-8 |
| EC Number | 700-167-6 |
| SMILES | CCOC(=O)CC(=O)C1=CC(=C(C(=C1F)F)F)F |
| Melting point | 41-44 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Ethyl 2,3,4,5-tetrafluorobenzoyl acetate is a notable chemical compound recognized for its unique structural attributes and diverse applications in both chemical synthesis and material science. This compound, characterized by its tetrafluorobenzoyl and acetate functional groups, presents interesting properties that contribute to its significance in various industrial and research contexts. The discovery of ethyl 2,3,4,5-tetrafluorobenzoyl acetate stems from the broader exploration of fluorinated aromatic compounds and their derivatives. Fluorinated compounds have long been of interest due to their distinctive chemical properties, such as enhanced stability, reactivity, and lipophilicity. The introduction of fluorine atoms into an aromatic ring can significantly alter the electronic and steric properties of the molecule, making it a valuable tool in organic synthesis and materials chemistry. Ethyl 2,3,4,5-tetrafluorobenzoyl acetate was developed as part of ongoing efforts to create highly reactive and versatile fluorinated intermediates. Its structure includes a benzoyl group with four fluorine atoms attached to the ring and an ester group linked to the benzoyl moiety. This arrangement endows the compound with unique reactivity patterns, making it suitable for a range of applications. In chemical synthesis, ethyl 2,3,4,5-tetrafluorobenzoyl acetate serves as a key intermediate in the preparation of various fluorinated compounds. The presence of the tetrafluorobenzoyl group introduces strong electron-withdrawing effects, which can facilitate nucleophilic substitution reactions and other transformations. This property is valuable for synthesizing complex molecules in pharmaceuticals and agrochemicals. Moreover, the compound's ester functionality is reactive towards nucleophiles, making it useful in esterification and hydrolysis reactions. These characteristics enable the creation of a wide array of fluorinated derivatives with tailored properties, which are essential for developing new materials and fine chemicals. In materials science, ethyl 2,3,4,5-tetrafluorobenzoyl acetate is employed in the synthesis of fluorinated polymers and materials. The inclusion of fluorine atoms in polymers can impart desirable properties such as enhanced chemical resistance, thermal stability, and low surface energy. These features make fluorinated polymers suitable for applications in coatings, adhesives, and electronic materials. The compound also plays a role in the development of advanced functional materials. For instance, it can be used to prepare fluorinated ligands for coordination chemistry, which are essential for designing new catalysts and materials with specific reactivity and selectivity. In summary, ethyl 2,3,4,5-tetrafluorobenzoyl acetate is a valuable chemical compound with significant applications in chemical synthesis and materials science. Its unique structural features and reactivity make it a versatile intermediate for creating fluorinated derivatives and advanced materials. References 2015. Synthesis, reactivity, and biological activity of 5-aminouracil and its derivatives. Molecular Diversity, 19(2). DOI: 10.1007/s11030-015-9595-1 2012. Features of synthesis and structure of ethyl (2Z)-3-hydroxy-(2,3,4,5-tetrafluorophenyl)-propyl-2-enoate. Russian Journal of General Chemistry, 82(1). DOI: 10.1134/s1070363212010197 2008. Condensation Reactions of Esters or Ortho Esters with Acidic Methylene Derivatives. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-032-00671 |
| Market Analysis Reports |