Online Database of Chemicals from Around the World
| Wuhan Hengheda Pharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hhdpharm.com | |||
 | +86 18086046872 | |||
 | hhdpharm@hotmail.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Zhengzhou Xlinebio Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xlinebio.com | |||
 | +86 (371) 6131-8675 | |||
 | +86 (371) 6131-8675 | |||
 | sales@xlinebio.com | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Xi'an Yinherb Bio-tech Co., Ltd | China | |||
|---|---|---|---|---|
 | yinherb.ecer.com | |||
 | +86 (29) 6333-2330 | |||
 | sales@yinherb-nutraceuticals.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Shaanxi Greenyo Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.greenyobio.com | |||
 | +86 (29) 8177-2657 | |||
 | sales05@greenyobio.com | |||
| Chemical distributor since 2017 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Salt of carboxylic acid ester and its derivatives |
|---|---|
| Name | 3-(2-Benzyl(methyl)aminoethyl)benzoic acid methyl ester hydrochloride |
| Synonyms | PRL 8-53; m-[2-(Benzylmethylamino)ethyl]benzoic acid methyl ester hydrochloride |
| Molecular Structure | 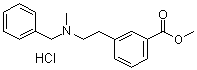 |
| Molecular Formula | C18H21NO2.HCl |
| Molecular Weight | 319.83 |
| CAS Registry Number | 51352-87-5 |
| SMILES | CN(CCC1=CC(=CC=C1)C(=O)OC)CC2=CC=CC=C2.Cl |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H312-H332 Details |
| Safety Statements | P261-P264-P280-P301+312-P302+352-P304+340-P321-P330-P362+364 Details |
| SDS | Available |
|
3-(2-Benzyl(methyl)aminoethyl)benzoic acid methyl ester hydrochloride is an organic compound that belongs to the class of substituted benzoic acid esters, functionalized with a tertiary amine side chain and isolated as a hydrochloride salt. Its structure features a benzoic acid core where the carboxylic acid is esterified to a methyl group, and the 3-position of the aromatic ring is substituted with a 2-(benzyl(methyl)amino)ethyl group. The hydrochloride salt form enhances the compound’s solubility and stability, particularly in aqueous environments. The molecule contains both lipophilic and hydrophilic components, with the aromatic ring and benzyl substituent providing hydrophobic character, while the protonated tertiary amine contributes to hydrophilicity under physiological conditions. The presence of the ester functional group suggests the molecule may undergo hydrolysis under enzymatic or acidic/basic conditions, yielding the corresponding carboxylic acid. Compounds of this general type are structurally related to local anesthetics or certain sympathomimetic agents, where aromatic esters and basic amines are commonly found. The tertiary amine motif is also reminiscent of structures seen in neurotransmitter analogs or transporter ligands. Depending on stereochemistry and specific substitutions, such molecules may interact with biological targets such as adrenergic, serotonergic, or dopaminergic systems, although this particular compound’s pharmacological profile would need to be defined through bioassays. As a hydrochloride salt, this compound is typically prepared by reacting the free base form with hydrogen chloride in an organic solvent, leading to precipitation of the crystalline hydrochloride. This salt form is often preferred for pharmaceutical candidates due to its improved handling characteristics. In synthetic chemistry, 3-(2-Benzyl(methyl)aminoethyl)benzoic acid methyl ester hydrochloride can serve as an intermediate for producing more complex bioactive molecules, particularly those targeting the central nervous system. Functional group modifications at the ester or amine positions offer routes for derivatization into amides, acids, quaternary ammonium salts, or other analogues with potentially altered bioactivity. References 1978. PRL-8-53: Enhanced learning and subsequent retention in humans as a result of low oral doses of new psychotropic agent. Psychopharmacology, 56(3). DOI: 10.1007/bf00432846 |
| Market Analysis Reports |