Online Database of Chemicals from Around the World
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Zhuyao Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | cn.pharmalego.com | |||
 | +86 (021) 5859-6383 | |||
 | marketing@pharmalego.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | 4-Bromo-2-methyl-2H-pyrazole-3-carboxylic acid methyl ester |
| Synonyms | Methyl 4-bromo-1-methyl-1H-pyrazole-5-carboxylate |
| Molecular Structure | 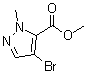 |
| Molecular Formula | C6H7BrN2O2 |
| Molecular Weight | 219.04 |
| CAS Registry Number | 514816-42-3 |
| EC Number | 836-336-7 |
| SMILES | CN1C(=C(C=N1)Br)C(=O)OC |
| Solubility | Slightly soluble (4.4 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.67±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| Index of Refraction | 1.590, Calc.* |
| Boiling point | 276.5±20.0 °C (760 Torr), Calc.* |
| Flash point | 121.0±21.8 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
4-Bromo-2-methyl-2H-pyrazole-3-carboxylic acid methyl ester is a chemical compound characterized by its pyrazole core, a five-membered heterocyclic structure that is widely used in organic synthesis and medicinal chemistry. The compound features a bromo group at the 4-position, a methyl group at the 2-position, and a methyl ester functionality attached to the carboxyl group at the 3-position of the pyrazole ring. This specific combination of substituents gives the molecule unique reactivity and potential for various applications. The discovery of pyrazole derivatives like 4-bromo-2-methyl-2H-pyrazole-3-carboxylic acid methyl ester can be traced back to the 19th century when pyrazole itself was first synthesized. Over the years, researchers have explored the incorporation of different functional groups at various positions of the pyrazole ring to create molecules with enhanced properties, such as increased biological activity or better solubility for pharmaceutical applications. The presence of a bromine atom is especially significant, as halogenated compounds often exhibit unique electronic properties that can influence their reactivity and interactions with biological targets. One of the primary applications of 4-bromo-2-methyl-2H-pyrazole-3-carboxylic acid methyl ester is in medicinal chemistry. Pyrazole derivatives have been found to possess a wide range of biological activities, including antibacterial, antifungal, anticancer, and anti-inflammatory properties. The incorporation of a bromine atom in this compound may enhance its lipophilicity and enable it to interact more effectively with hydrophobic regions of biological targets, such as proteins or enzymes. Additionally, the methyl ester group can provide the compound with improved solubility, facilitating its absorption and bioavailability in therapeutic contexts. In drug development, compounds like 4-bromo-2-methyl-2H-pyrazole-3-carboxylic acid methyl ester are often synthesized as part of high-throughput screening efforts to identify potential leads for novel drugs. Pyrazole-based molecules are particularly valuable as inhibitors of protein kinases, enzymes that play crucial roles in cell signaling and cancer progression. The bromine atom and the ester group in this compound may contribute to its ability to form specific interactions with the active sites of these enzymes, making it a candidate for further optimization in the search for targeted therapies. Beyond medicinal chemistry, 4-bromo-2-methyl-2H-pyrazole-3-carboxylic acid methyl ester also finds applications in organic synthesis as an intermediate in the preparation of more complex pyrazole-containing compounds. The versatility of the pyrazole ring allows for the construction of a wide variety of derivatives with tailored properties, such as fluorescent dyes, agrochemicals, and materials for electronics. The bromine atom, in particular, can participate in cross-coupling reactions, making the compound a valuable building block for the synthesis of larger, more complex molecules. In materials science, pyrazole derivatives are increasingly being explored for their potential as organic semiconductors, light-emitting materials, and sensors. The functionalization of the pyrazole ring with different substituents, such as bromine and ester groups, allows for fine-tuning of the molecule’s electronic properties. This enables the development of materials with desirable optical and electrical characteristics for use in organic light-emitting diodes (OLEDs), organic solar cells, and other advanced electronic devices. In summary, 4-bromo-2-methyl-2H-pyrazole-3-carboxylic acid methyl ester is a compound with significant potential in medicinal chemistry, organic synthesis, and materials science. Its structure, characterized by a brominated pyrazole ring and an ester functional group, positions it as a valuable building block for drug discovery and the development of novel materials. Ongoing research into pyrazole derivatives like this one continues to unlock new applications in a range of fields, from healthcare to electronics. References 2023. Multimodal action of KRP203 on phosphoinositide kinases in vitro and in cells. Biochemical and Biophysical Research Communications, 678. DOI: 10.1016/j.bbrc.2023.08.050 2022. A quantitative high-throughput screen identifies compounds that lower expression of the SCA2-and ALS-associated gene ATXN2. The Journal of biological chemistry, 298(8). DOI: 10.1016/j.jbc.2022.102228 2019. BioTransformer: a comprehensive computational tool for small molecule metabolism prediction and metabolite identification. Journal of Cheminformatics, 11(1). DOI: 10.1186/s13321-018-0324-5 |
| Market Analysis Reports |