Online Database of Chemicals from Around the World
| YURUI (Shanghai) Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.riyngroup.com | |||
 | +86 (21) 3319-1321 ex 816 +86 13032131612 | |||
 | +86 (21) 6085-3441 | |||
 | yu@riyngroup.com info@riyngroup.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Luminescence Technology Corp. | Taiwan | |||
|---|---|---|---|---|
 | www.lumtec.com.tw | |||
 | +886 (2) 2697-5600 | |||
 | +886 (2) 2697-5601 | |||
 | sales@lumtec.com.tw | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Fluorenes |
|---|---|
| Name | 2,2',7,7'-Tetrakis(di-p-tolylamino)spiro-9,9'-bifluorene |
| Synonyms | 2,2',7,7'-Tetrakis(N,N-ditolylamino)-spiro-9,9'-bifluorene; 2,2',7,7'-Tetrakis(di-4-methylphenylamino)-9,9'-spirobifluorene; HIM; HIM 1; Spiro-TTB |
| Molecular Structure | 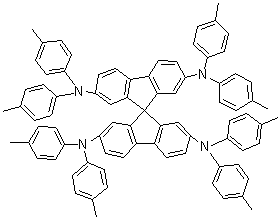 |
| Molecular Formula | C81H68N4 |
| Molecular Weight | 1097.43 |
| CAS Registry Number | 515834-67-0 |
| SMILES | CC1=CC=C(C=C1)N(C2=CC=C(C=C2)C)C3=CC4=C(C=C3)C5=C(C46C7=C(C=CC(=C7)N(C8=CC=C(C=C8)C)C9=CC=C(C=C9)C)C1=C6C=C(C=C1)N(C1=CC=C(C=C1)C)C1=CC=C(C=C1)C)C=C(C=C5)N(C1=CC=C(C=C1)C)C1=CC=C(C=C1)C |
| Solubility | Insoluble (2.6E-14 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.28±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2014 ACD/Labs) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319 Details |
| Safety Statements | P264-P280-P302+P352-P337+P313-P305+P351+P338-P362+P364-P332+P313 Details |
| SDS | Available |
|
2,2',7,7'-Tetrakis(di-p-tolylamino)spiro-9,9'-bifluorene is an organic compound with significant applications in the fields of material science and electronics. This compound belongs to a class of organic semiconductors known as spirobifluorenes, which are characterized by their unique molecular structure, where two fluorene units are connected through a spiro linkage. The structure of 2,2',7,7'-Tetrakis(di-p-tolylamino)spiro-9,9'-bifluorene consists of a central spirobifluorene core with four di-p-tolylamino groups attached to the 2 and 7 positions of the fluorene units. These amino groups contribute to the compound's electronic properties, making it an effective material for optoelectronic applications. The discovery of 2,2',7,7'-Tetrakis(di-p-tolylamino)spiro-9,9'-bifluorene can be traced back to research in the early 21st century, which focused on the development of organic materials with high charge transport capabilities. Researchers sought to design organic compounds that could be used in organic light-emitting diodes (OLEDs), solar cells, and field-effect transistors (FETs). The spirobifluorene structure emerged as a promising candidate due to its high thermal stability, rigid molecular framework, and ability to facilitate efficient charge transport. One of the key applications of 2,2',7,7'-Tetrakis(di-p-tolylamino)spiro-9,9'-bifluorene is in the field of organic electronics, particularly in the development of organic light-emitting diodes (OLEDs). OLEDs are used in a variety of devices, including flat-panel displays, televisions, and lighting panels. The di-p-tolylamino groups on the spirobifluorene core improve the compound's ability to transport charge, which is essential for efficient light emission in OLEDs. This organic semiconductor is known to enhance the performance of OLED devices by increasing their brightness, efficiency, and stability. In addition to its use in OLEDs, 2,2',7,7'-Tetrakis(di-p-tolylamino)spiro-9,9'-bifluorene has potential applications in organic photovoltaic (OPV) cells. OPVs are a type of solar cell that use organic materials to convert sunlight into electricity. The spirobifluorene structure, with its high electronic mobility and stability, makes it an ideal candidate for use in the active layers of OPVs. By incorporating 2,2',7,7'-Tetrakis(di-p-tolylamino)spiro-9,9'-bifluorene into these devices, researchers aim to improve their efficiency and long-term stability. The compound is also being explored for use in organic field-effect transistors (OFETs), which are essential components in flexible electronics, sensors, and memory devices. The high charge mobility of 2,2',7,7'-Tetrakis(di-p-tolylamino)spiro-9,9'-bifluorene makes it suitable for use in the semiconducting layers of OFETs, where efficient charge transport is critical to the performance of these devices. Moreover, due to its unique molecular structure, 2,2',7,7'-Tetrakis(di-p-tolylamino)spiro-9,9'-bifluorene is also being investigated for its potential in other areas of organic electronics, such as sensors, memory devices, and flexible electronics. Its ability to form stable thin films and its compatibility with solution-processing techniques make it an attractive material for the development of large-area, low-cost electronic devices. In conclusion, 2,2',7,7'-Tetrakis(di-p-tolylamino)spiro-9,9'-bifluorene is a promising organic semiconductor with significant applications in the fields of organic electronics, particularly in OLEDs, OPVs, and OFETs. Its high charge transport properties, stability, and ease of processing make it an attractive material for the next generation of flexible, lightweight, and efficient electronic devices. References 2023. OLED-on-CMOS probes for single-neuron optogenetics. Nature Electronics, 6(9). DOI: 10.1038/s41928-023-01032-9 2023. Optogenetic stimulation probes with single-neuron resolution based on organic LEDs monolithically integrated on CMOS. Nature Electronics, 6(8). DOI: 10.1038/s41928-023-01013-y 2019. Flexible organic light-emitting diodes for antimicrobial photodynamic therapy. npj Flexible Electronics, 3(1). DOI: 10.1038/s41528-019-0058-0 |
| Market Analysis Reports |