Online Database of Chemicals from Around the World
| Xiamen Pioneer Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xm-pioneer.com | |||
 | +86 (592) 392-2797 | |||
 | +86 (592) 377-6077 | |||
 | xmpt@xm-pioneer.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Dishman Pharmaceuticals & Chemicals Limited | USA | |||
|---|---|---|---|---|
 | www.dishmangroup.com | |||
 | +1 (732) 560-4300 | |||
 | +1 (732) 560-4343 | |||
 | bhaveshoza@dishmangroup.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Medilink Pharmachem | India | |||
|---|---|---|---|---|
 | www.medilinkpharma.com | |||
 | +91 (79) 3007-0133 | |||
 | +91 (79) 4007-4175 | |||
 | exports@medilinkpharma.com | |||
| Chemical distributor since 1996 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Guizhou Jianye Yongrong Technology Co., Ltd. (Changzhou Huadong Chemical Research Institute) | China | |||
|---|---|---|---|---|
 | www.huadongchem.com | |||
 | +86 +13851563211 | |||
 | +86 (519) 8261-6386 | |||
 | sales@huadongchem.com | |||
 | WeChat: +13851563211 | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Zhejiang Xianju Application Chemical Research Institute | China | |||
|---|---|---|---|---|
 | www.chemptc.com | |||
 | +86 (576) 8768-8839 8760-1639 8768-9177 | |||
 | +86 (576) 8768-8808 | |||
 | lucychen@chemrole.com | |||
| Chemical manufacturer since 1993 | ||||
| Jintan Xinan Chemical Institue | China | |||
|---|---|---|---|---|
 | www.xinan-chem.com | |||
 | +86 (519) 8261-8828 8261-8838 8261-1598 | |||
 | +86 (519) 8261-8836 / 8261-8829 | |||
 | sales@xinan-chem.com | |||
| Chemical manufacturer since 1994 | ||||
| Shanghai Cainorise Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cainorise.com.cn | |||
 | +86 (21) 3408-0216 2950-8524 +86 15921434758 | |||
 | sales@cainorise.com.cn | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Amine salt (ammonium salt) |
|---|---|
| Name | Benzyltriethylammonium bromide |
| Molecular Structure | 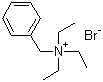 |
| Molecular Formula | C13H22N.Br |
| Molecular Weight | 272.23 |
| CAS Registry Number | 5197-95-5 |
| EC Number | 225-986-1 |
| SMILES | CC[N+](CC)(CC)CC1=CC=CC=C1.[Br-] |
| Melting point | 193-195 °C (Expl.) |
|---|---|
| Water solubility | soluble |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| Transport Information | UN 2811 | ||||||||||||
| SDS | Available | ||||||||||||
|
Benzyltriethylammonium bromide is a quaternary ammonium salt composed of a benzyl group attached to a nitrogen atom, which is further bonded to three ethyl groups, and a bromide counterion. It is a member of the family of alkylammonium salts, which are widely used in organic chemistry and industrial applications. The compound is typically synthesized by reacting benzyl chloride with triethylamine, a well-established method that leads to the formation of benzyltriethylammonium bromide as a salt with a bromide anion. This compound is primarily utilized in phase transfer catalysis, a technique where it facilitates the transfer of reactants between two immiscible phases, typically an aqueous phase and an organic phase. In these reactions, the benzyltriethylammonium bromide serves as a phase transfer agent by solubilizing polar or ionic compounds in organic solvents, allowing them to react with other substances in the organic phase. This makes it particularly valuable in nucleophilic substitution reactions or other organic transformations where reactants in an aqueous phase need to interact with reactants in an organic phase. Beyond phase transfer catalysis, benzyltriethylammonium bromide is employed in the synthesis of various organic compounds. It acts as a reagent in the formation of amines, amides, and other nitrogen-containing organic molecules. The presence of the benzyl and ethyl groups on the ammonium ion imparts specific steric and electronic effects that make it useful for certain synthetic applications. These properties are leveraged in the development of complex molecules in medicinal chemistry and industrial organic synthesis. Additionally, benzyltriethylammonium bromide has applications in ion-exchange processes, where it can serve as a source of the triethylammonium ion in various chemical reactions. It is also used as a surfactant and emulsifying agent in various chemical formulations, such as detergents, coatings, and cleaning agents. Its amphiphilic nature, due to the hydrophilic ammonium group and hydrophobic alkyl chains, allows it to stabilize emulsions and suspensions effectively. In terms of safety, as a quaternary ammonium compound, benzyltriethylammonium bromide must be handled with care. It is important to follow standard safety protocols, such as wearing appropriate protective equipment and handling the substance in a well-ventilated area, to minimize the risk of exposure. Although it is generally considered to be of low toxicity, prolonged exposure or improper handling could lead to irritation or other adverse health effects. In conclusion, benzyltriethylammonium bromide is a valuable chemical compound widely used in organic synthesis, phase transfer catalysis, and various industrial applications. Its ability to facilitate the transfer of reactants between different phases and its use in the synthesis of nitrogen-containing compounds make it an important reagent in both research and industrial settings. Its broad range of applications and continued use in chemical processes reflect its significance in modern chemistry. References 2023 Synthesis of Atropisomeric Benzoxazocines Based on Etherification Products of N-[2-(Cycloalk-1-en-1-yl)phenyl]sulfonamides with Ethylene Chlorohydrin. Russian Journal of Organic Chemistry, 1, 1. DOI: 10.1134/s1070428023120138 2023 Synthesis of Thia- and Selenodiazoles Based on Acetosyringone and Its Glycosides. Russian Journal of General Chemistry, 1, 1. DOI: 10.1134/s1070363223140013 2022 Metal-mediated reactions of bromoform with electron-rich and electron-deficient carbon-carbon and carbon-hetero atom multiple bonds. Journal of Chemical Sciences, 1, 1. DOI: 10.1007/s12039-022-02075-9 |
| Market Analysis Reports |