Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Classification | Chemical reagent >> Organic reagent >> Amine salt (ammonium salt) |
|---|---|
| Name | 6-(Bromomethyl)-2,4-pteridinediamine hydrobromide |
| Molecular Structure | 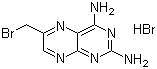 |
| Molecular Formula | C7H72N6.HBr |
| Molecular Weight | 335.99 |
| CAS Registry Number | 52853-40-4 |
| EC Number | 692-499-2 |
| SMILES | C1=C(N=C2C(=NC(=NC2=N1)N)N)CBr.Br |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
6-(Bromomethyl)-2,4-pteridinediamine hydrobromide is a chemical compound primarily used in the field of medicinal chemistry. It consists of a pteridine ring system, a heterocyclic structure that plays a crucial role in various biological processes, and a bromomethyl group attached to the pteridine nitrogen. The compound also contains hydrobromide salt, which is often used to stabilize its molecular structure. This substance has been explored for its potential applications in the synthesis of biologically active molecules, including those with anticancer and anti-inflammatory properties. The discovery of 6-(Bromomethyl)-2,4-pteridinediamine hydrobromide is linked to the broader field of pteridine derivatives, which have been studied for their wide range of biological activities. Pteridines, including derivatives such as folic acid and methotrexate, are well-known for their involvement in cellular metabolism, particularly in the synthesis of nucleotides and amino acids. The modification of the pteridine structure, especially with functional groups like bromomethyl, has expanded the range of potential biological targets and enhanced its pharmaceutical applications. One of the major applications of 6-(Bromomethyl)-2,4-pteridinediamine hydrobromide is in the development of anticancer agents. The pteridine ring structure is known to interact with enzymes involved in DNA synthesis, making it a valuable scaffold for drug development aimed at interfering with cell division. In particular, compounds containing a bromomethyl group can undergo nucleophilic substitution reactions with biological targets, facilitating their binding to proteins or nucleic acids, which could ultimately lead to the inhibition of cancer cell proliferation. The hydrobromide form of the compound helps to increase its solubility and stability, making it more suitable for use in pharmaceutical formulations. In addition to its anticancer potential, 6-(Bromomethyl)-2,4-pteridinediamine hydrobromide has been investigated for its anti-inflammatory properties. Pteridine derivatives have been shown to exhibit activity against various inflammatory mediators, and the introduction of the bromomethyl group may enhance this activity. The compound has been studied in vitro and in animal models for its ability to reduce inflammation and alleviate symptoms associated with conditions such as rheumatoid arthritis and other autoimmune disorders. Synthetically, 6-(Bromomethyl)-2,4-pteridinediamine hydrobromide is typically prepared through the bromomethylation of a 2,4-pteridinediamine derivative. The process involves the reaction of the pteridine compound with a bromomethylating agent, such as formaldehyde and hydrogen bromide, which introduces the bromomethyl group to the pteridine ring. The resulting hydrobromide salt is isolated through crystallization, providing a stable, soluble form of the compound suitable for further research and drug development. In conclusion, 6-(Bromomethyl)-2,4-pteridinediamine hydrobromide is a valuable chemical substance with potential applications in drug discovery, particularly in the development of anticancer and anti-inflammatory agents. Its unique pteridine structure, combined with the bromomethyl and hydrobromide functional groups, makes it a promising candidate for further research into novel therapeutic compounds. References 2003. Methotrexate. Pharmaceutical Substances. URL: https://pharmaceutical-substances.thieme.com/ps/search-results?docUri=KD-13-0098 1983. A Synthetic Approach to Poly-γ-Glutamyl Analogs of Methotrexate. Advances in Experimental Medicine and Biology. DOI: 10.1007/978-1-4757-5241-0_9 |
| Market Analysis Reports |