Online Database of Chemicals from Around the World
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2010 | ||||
| Classification | Natural product >> Natural phenols |
|---|---|
| Name | 6-Cyano-2-naphthol |
| Synonyms | 6-hydroxynaphthalene-2-carbonitrile |
| Molecular Structure | 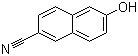 |
| Molecular Formula | C11H7NO |
| Molecular Weight | 169.18 |
| CAS Registry Number | 52927-22-7 |
| EC Number | 628-663-7 |
| SMILES | C1=CC2=C(C=CC(=C2)O)C=C1C#N |
| Density | 1.3±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 165.5-170.5 °C (Expl.) |
| Index of Refraction | 1.692, Calc.* |
| Boiling Point | 383.1±15.0 °C (760 mmHg), Calc.* |
| Flash Point | 185.5±20.4 °C, Calc.* |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
6-Cyano-2-naphthol is an organic compound that combines the properties of both a naphthalene ring structure and a cyano group. This compound is characterized by a hydroxyl group (-OH) attached to the second carbon of the naphthalene ring and a cyano group (-CN) attached to the sixth carbon. The presence of the cyano group introduces strong electron-withdrawing characteristics, which affects the compound's reactivity, making it a useful intermediate in organic synthesis. The discovery of 6-cyano-2-naphthol can be attributed to the need for versatile building blocks in the development of various chemicals, particularly those with applications in pharmaceuticals, agrochemicals, and dyes. The compound’s structure, featuring a naphthalene core with functional groups that can participate in further chemical transformations, made it an attractive candidate for exploration in synthetic chemistry. Researchers found that the cyano group significantly influences the compound's electronic properties, which is essential for its applications. The synthesis of 6-cyano-2-naphthol typically involves several steps that include the functionalization of naphthalene. One common synthetic route is the nitration of naphthalene, followed by selective reduction and subsequent introduction of the cyano group. Various methods, such as nucleophilic substitution and electrophilic aromatic substitution, can be employed depending on the specific reaction conditions and desired yield. The process requires careful control to achieve high selectivity and purity in the final product. 6-Cyano-2-naphthol has several important applications in organic synthesis and materials science. One of its primary uses is as an intermediate in the synthesis of various pharmaceuticals, including compounds that act on the central nervous system, antimicrobial agents, and anticancer drugs. The compound's ability to undergo nucleophilic substitution reactions makes it valuable in the creation of complex molecules with high selectivity. Additionally, 6-cyano-2-naphthol serves as an intermediate in the production of dyes and pigments. Its structure allows it to be incorporated into more complex dye molecules, providing vibrant colors and stability, which is critical in industries such as textiles, plastics, and printing. The compound is also studied for its potential in the development of materials with specialized optical properties, such as organic semiconductors and photovoltaic devices. In conclusion, 6-cyano-2-naphthol is a versatile and important chemical intermediate with a range of applications in both industrial and pharmaceutical chemistry. Its ability to undergo various chemical transformations makes it a valuable tool for the synthesis of complex compounds. Continued research into its properties and applications holds promise for further innovations in organic materials and drug development. References 2018. C-Glycosylation of Substituted β-Naphthols with Trichloroacetimidate Glycosyl Donors. Synthesis, 50(3). DOI: 10.1055/s-0036-1591746 2011. Simple and Efficient One-Step Conversion of Benzonitrile into Methylarene under Mild Conditions. Synlett, 2011(14). DOI: 10.1055/s-0030-1261164 |
| Market Analysis Reports |