Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organotin |
|---|---|
| Name | Stannous methanesulfonate |
| Synonyms | Methanesulfonic acid tin(2+) salt; Tin(II) methanesulfonate |
| Molecular Structure | 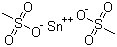 |
| Molecular Formula | Sn(CH3O3S)2 |
| Molecular Weight | 308.91 |
| CAS Registry Number | 53408-94-9 |
| EC Number | 610-996-4 |
| SMILES | CS(=O)(=O)[O-].CS(=O)(=O)[O-].[Sn+2] |
| Density | 1.55 g/mL (Expl.) |
|---|---|
| Melting point | -27 °C (Expl.) |
| Refractive index | 1.444 |
| Hazard Symbols |    GHS05;GHS07;GHS09 Danger Details GHS05;GHS07;GHS09 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H314-H317-H318-H411 Details | ||||||||||||||||||||
| Safety Statements | P260-P261-P264-P264+P265-P270-P272-P273-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P330-P333+P317-P362+P364-P363-P391-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| Transport Information | UN 3265 | ||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Stannous methanesulfonate, also known as tin(II) methanesulfonate, is a chemical compound consisting of tin in the +2 oxidation state and the methanesulfonate anion. This compound has found its place in various industrial and chemical applications, particularly due to its unique properties as a reducing agent and its role in the synthesis of other chemical compounds. The discovery of stannous methanesulfonate is rooted in the broader exploration of tin compounds and their reactions. Tin has long been recognized for its ability to undergo redox reactions, and the development of stannous salts, including stannous methanesulfonate, has been a significant part of advancing its use in organic chemistry. The methanesulfonate group (–CH3SO3) is known for its stability and ability to form strong bonds with metal ions, making stannous methanesulfonate a useful reagent in several chemical processes. Stannous methanesulfonate is primarily used as a reducing agent in chemical synthesis. It plays a crucial role in organic reactions, particularly in the reduction of various functional groups, such as carbonyl compounds, nitriles, and imines. The compound’s ability to transfer electrons makes it a valuable tool for chemists who require mild reduction conditions without the need for more hazardous or expensive reagents. In this regard, stannous methanesulfonate is often preferred due to its manageable toxicity and high efficiency in many reduction reactions. In addition to its role as a reducing agent, stannous methanesulfonate is used in the synthesis of other tin-based compounds. For example, it is involved in the preparation of organotin compounds, which are widely used as stabilizers in plastics, pesticides, and other industrial applications. The versatile chemistry of stannous methanesulfonate allows it to participate in various organic transformations, including nucleophilic substitution and coupling reactions, further enhancing its utility in synthetic chemistry. The compound is also utilized in the preparation of certain catalytic systems. In the field of catalysis, stannous methanesulfonate has been explored as a catalyst in certain polymerization reactions and in the production of specialty materials. Its relatively low cost and ease of handling make it an attractive option for industrial applications where cost-effective and efficient processes are critical. In the pharmaceutical and fine chemicals industries, stannous methanesulfonate’s reducing properties are used in the synthesis of various intermediates, which can be further modified to create complex molecules. Its ability to selectively reduce certain bonds while leaving others intact is valuable in the synthesis of molecules with specific functional groups, which is essential in the development of new drugs and materials. While its primary applications lie in chemical synthesis and catalysis, stannous methanesulfonate’s use is not without limitations. The compound can be sensitive to air and moisture, which may affect its stability. However, when handled correctly, it remains an efficient and versatile reagent that offers significant advantages in a range of chemical processes. In conclusion, stannous methanesulfonate is an important chemical compound with diverse applications in synthetic chemistry and industrial processes. Its role as a reducing agent, its use in organotin chemistry, and its involvement in catalytic systems make it a valuable tool for researchers and industries alike. Continued exploration of its properties and reactions will likely reveal even more applications in the years to come. References none |
| Market Analysis Reports |