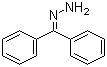
Benzophenone hydrazone is an organic compound derived from the condensation reaction of benzophenone and hydrazine. Its chemical formula is C13H12N2O, and it belongs to the class of hydrazones, which are characterized by the presence of a hydrazone functional group (-C=N-NH2). The discovery of benzophenone hydrazone is part of a broader exploration of hydrazone derivatives, which began in the late 19th century as chemists sought to develop new compounds for various applications.
The synthesis of benzophenone hydrazone was first reported in the early 20th century when researchers discovered the ability of hydrazine to react with carbonyl compounds, leading to the formation of hydrazones. This reaction is known for its simplicity and efficiency, allowing for the generation of a wide range of hydrazone derivatives. The unique structural features of benzophenone hydrazone, including the phenyl groups and the hydrazone linkage, contribute to its chemical reactivity and potential applications in diverse fields.
One of the primary applications of benzophenone hydrazone is in the realm of organic synthesis, where it serves as an important intermediate in the production of various compounds. Its ability to undergo further chemical transformations, such as oxidation, reduction, and substitution reactions, makes it a valuable building block in the synthesis of pharmaceuticals, agrochemicals, and other organic materials. For instance, benzophenone hydrazone can be converted into more complex molecules, including azo compounds and heterocyclic structures, which have significant importance in medicinal chemistry.
In addition to its role in organic synthesis, benzophenone hydrazone has been explored for its potential applications in the field of photochemistry. The compound exhibits UV-absorbing properties, making it useful in the formulation of sunscreen and cosmetic products. By incorporating benzophenone hydrazone into formulations, manufacturers can enhance the UV protection offered by their products, thereby improving their efficacy and consumer appeal. This application is particularly relevant in an era where skin protection from UV radiation is increasingly emphasized for preventing skin damage and reducing the risk of skin cancer.
Benzophenone hydrazone has also garnered interest in the field of materials science, particularly in the development of polymeric materials. Its ability to undergo cross-linking reactions when exposed to UV light allows for the creation of photo-cross-linkable resins, which find applications in coatings, adhesives, and 3D printing technologies. The incorporation of benzophenone hydrazone into these materials enhances their mechanical properties, durability, and resistance to environmental factors, making them suitable for various industrial applications.
Moreover, benzophenone hydrazone has been investigated for its biological activities, particularly in the context of drug discovery. Preliminary studies have suggested that certain hydrazone derivatives may exhibit antimicrobial, anticancer, and anti-inflammatory properties. This has led researchers to explore the structure-activity relationship of benzophenone hydrazone and its derivatives, aiming to identify compounds with enhanced biological efficacy for potential therapeutic applications.
Despite its promising applications, the use of benzophenone hydrazone requires careful consideration of safety and regulatory aspects. As with many organic compounds, appropriate handling and disposal measures must be observed to mitigate any potential health and environmental risks associated with its use.
In summary, benzophenone hydrazone is a significant compound with a rich history of discovery and diverse applications in organic synthesis, photochemistry, materials science, and drug discovery. Its versatile chemical properties and reactivity continue to make it a valuable subject of study, highlighting its importance across multiple scientific disciplines.
References
2024. Synthesis of N-(iminyl)pyridinium salts from hydrazones by the Zincke reaction. Russian Chemical Bulletin, 73(5).
DOI: 10.1007/s11172-024-4246-2
2023. First Diels-Alder reaction of a N-nitrogen-substituted iminium ion as dienophile furnishing N-amino-substituted 1,2,5,6-tetrahydropyridines. Monatshefte für Chemie - Chemical Monthly, 154(4).
DOI: 10.1007/s00706-023-03054-7
|











































































 GHS06;GHS07;GHS09 Danger Details
GHS06;GHS07;GHS09 Danger Details