Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Chemical reagent >> Organic reagent >> Carboxylic anhydride |
|---|---|
| Name | 1,2,4-Cyclohexanetricarboxylic anhydride |
| Synonyms | 4-Carboxyhexahydrophthalic anhydride; Cyclohexane-1,2,4-tricarboxylic acid anhydride; Cyclohexane-1,3,4-tricarboxylic acid 3,4-anhydride; H-TMAn; H-TMAn-s; Hydrogenated trimellitic anhydride |
| Molecular Structure | 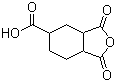 |
| Molecular Formula | C9H10O5 |
| Molecular Weight | 198.17 |
| CAS Registry Number | 53611-01-1 |
| EC Number | 800-369-5 |
| SMILES | C1CC2C(CC1C(=O)O)C(=O)OC2=O |
| Density | 1.446 |
|---|---|
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H317-H318-H334 Details | ||||||||||||||||
| Safety Statements | P261-P272-P280-P284-P302+P352-P305+P351+P338 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
1,2,4-Cyclohexanetricarboxylic anhydride, often referred to as trimellitic anhydride, is a versatile chemical compound with a broad range of applications in various industries. This anhydride derivative is produced by the dehydration of trimellitic acid and has the molecular formula C9H6O3. It consists of a cyclohexane ring with three carboxyl groups, each bonded to a carbon atom, which forms the anhydride structure. The compound is typically a white to light yellow solid and is known for its reactivity and ability to form ester bonds with alcohols. The discovery of 1,2,4-cyclohexanetricarboxylic anhydride can be traced back to the early 20th century, when chemists began studying the properties and reactions of organic anhydrides. Its precursor, trimellitic acid, was known to have a variety of industrial uses, but its transformation into the anhydride opened up new possibilities for its application, particularly in polymer chemistry and coatings. Today, 1,2,4-cyclohexanetricarboxylic anhydride is considered an important industrial chemical, finding use in the manufacture of resins, plastics, and other specialized materials. One of the primary applications of 1,2,4-cyclohexanetricarboxylic anhydride is in the production of polyimides, which are high-performance polymers known for their thermal stability and electrical insulating properties. These polymers are essential in the aerospace, automotive, and electronics industries. The compound acts as a key monomer or reactant in the synthesis of polyimide resins, which are then used to create films, coatings, and composites that can withstand extreme temperatures and harsh environmental conditions. In addition to its role in polyimide production, 1,2,4-cyclohexanetricarboxylic anhydride is used in the manufacture of plasticizers, which are additives that enhance the flexibility and durability of plastics. The compound's ability to form ester linkages with alcohols makes it an ideal building block for plasticizers used in the production of flexible materials such as PVC, rubber, and other synthetic polymers. Furthermore, this anhydride is used in the synthesis of special-purpose chemicals, including surface-active agents, corrosion inhibitors, and curing agents for various materials. Its reactivity with alcohols and amines allows for the creation of a wide range of chemical derivatives, which can be tailored for specific applications in the coatings, adhesives, and automotive industries. References none |
| Market Analysis Reports |