Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Biocompounds Pharmaceutical Inc. | China | |||
|---|---|---|---|---|
 | www.biocompounds.com | |||
 | +86 (21) 5768-7675 | |||
 | +86 (21) 5768-7169 | |||
 | sales@biocompounds.com info@biocompounds.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Nanjing Ange Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.angepharm.com | |||
 | +86 (25) 8621-5454 | |||
 | +86 (25) 8650-1191 | |||
 | sales@angepharm.com | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Fujian South Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.southpharma.com | |||
 | +86 (598) 286-0412 +86 13509335186 | |||
 | +86 (598) 286-0416 | |||
 | sales@southpharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Manus Aktteva | India | |||
|---|---|---|---|---|
 | www.manusaktteva.in | |||
 | +91 (79) 6512-3395 | |||
 | +91 (79) 2646-3395 | |||
 | products@manusakttevabiopharma.in | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hangzhou Rongda Pharm & Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rdpharm.com | |||
 | +86 (571) 8808-8387 8808-6013 | |||
 | +86 (571) 8808-8387ex 802 | |||
 | sales@rdpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Lianyungang United Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lyg-china.com | |||
 | +86 (518) 8569-2709 | |||
 | +86 (518) 8501-5182 | |||
 | service@lyg-china.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Shanghai Growingchem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.growingchem.com | |||
 | +86 (21) 5080-0795 | |||
 | +86 (21) 6496-3699 | |||
 | sales@growingchem.com syzhang1983@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Changzhou Sunchem Pharmaceutical Chemical Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunkechem.com | |||
 | +86 (519) 8519-5575 8519-5528 8519-5565 | |||
 | +86 (519) 8519-5585 | |||
 | info@sunkechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| KKyemistry (India) Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.kyemistryindia.com | |||
 | +91 (40) 2351-0950 | |||
 | +91 (40) 2351-0667 | |||
 | info@kyemistryindia.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Golden Promise Industry Int'l Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.goldenpromiseindustry.com | |||
 | +86 15065329263 | |||
 | goldenpromise@163.com | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Qichuang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qc-chemical.com | |||
 | +86 (571) 8893-5129 | |||
 | +86 (571) 8825-0182 | |||
 | david@qc-chemical.com sales@qc-chemical.com davidw0828@gmail.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Trignokem International | India | |||
|---|---|---|---|---|
 | www.trignokem.com | |||
 | +91 (40) 2717-0709 | |||
 | info@trignokem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Rochi Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rochipharma.com | |||
 | +86 (21) 3875-1876 +86 15000076078 | |||
 | +86 (21) 5027-5764 | |||
 | info@rochipharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Mayue Longteng Technology Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lantu-tech.com | |||
 | +86 15171477138 | |||
 | helen.yan@lantu-tech.com | |||
 | QQ Chat | |||
 | WeChat: 15171477138 | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Eburon Organics bvba | Belgium | |||
|---|---|---|---|---|
 | www.eburon-organics.com | |||
 | +32 (51) 335-068 | |||
 | +32 (51) 335-064 | |||
 | sales@eburon-organics.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Pharmaceutical intermediate >> API intermediate |
|---|---|
| Name | 3-Aminophenylacetylene |
| Synonyms | 3-Ethynylaniline |
| Molecular Structure | 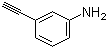 |
| Molecular Formula | C8H7N |
| Molecular Weight | 117.15 |
| CAS Registry Number | 54060-30-9 |
| EC Number | 258-944-6 |
| SMILES | C#CC1=CC(=CC=C1)N |
| Density | 1.1±0.1 g/cm3 Calc.*, 1.12 g/mL (Expl.) |
|---|---|
| Melting point | 27 °C (Expl.) |
| Boiling point | 240.5±23.0 °C 760 mmHg (Calc.)*, 282.3 °C (Expl.) |
| Flash point | 111.0±17.9 °C (Calc.)*, 59 °C (Expl.) |
| Index of refraction | 1.59 (Calc.)*, 1.617 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS02;GHS07 Warning Details GHS02;GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H315-H319-H335 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P261-P264-P264+P265-P271-P280-P302+P352-P303+P361+P353-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1993 | ||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
3-Aminophenylacetylene is an organic compound consisting of a phenyl ring substituted with an amino group at the meta-position (position 3) and an ethynyl group (-C≡CH) directly bonded to the ring. Its molecular structure can be described as a derivative of phenylacetylene, where the amine substituent is introduced at the 3-position, resulting in a compound with both nucleophilic and electrophilic reactive sites. The synthesis of 3-aminophenylacetylene typically involves functional group transformation of 3-nitrophenylacetylene, where the nitro group is reduced to a primary amine using standard reducing agents such as palladium on carbon with hydrogen, or by chemical reduction with tin(II) chloride or iron in acidic conditions. The acetylene group, being terminal and relatively reactive, allows for straightforward synthetic modifications, such as Sonogashira couplings, cycloadditions, or coordination to metal centers. 3-Aminophenylacetylene is of interest in various areas of chemical research, especially in the synthesis of conjugated systems and functional materials. The ethynyl group provides a linear extension to the aromatic system, enabling the formation of extended π-conjugated frameworks useful in the design of organic electronic materials, such as conductive polymers, light-emitting diodes, and photovoltaic materials. The amino group, being electron-donating, can influence the electronic properties of the molecule, making it suitable for tuning charge transport or light absorption characteristics. In the field of medicinal chemistry, aromatic acetylenes bearing amino groups have been studied as building blocks for the development of kinase inhibitors, enzyme modulators, and bioactive heterocycles. The ethynyl group serves as a versatile handle for further chemical elaboration, including the formation of heteroaromatic systems such as indoles or triazoles, especially through cycloaddition reactions like the Huisgen 1,3-dipolar cycloaddition. Additionally, the amino group can participate in hydrogen bonding or serve as a site for conjugation to larger biomolecules or polymers. The dual reactivity of 3-aminophenylacetylene allows it to serve as a bifunctional monomer in polymer chemistry. It can undergo polymerization reactions involving either the amino or ethynyl groups, leading to materials with diverse mechanical and optical properties. This makes it a valuable component in the design of specialty polymers, adhesives, or coatings with tailored performance characteristics. Analytically, derivatives of 3-aminophenylacetylene are used as intermediates in the synthesis of fluorescent dyes or molecular probes. The rigid and planar structure formed by conjugated systems containing phenylacetylene moieties allows for strong fluorescence and favorable binding to biomolecules. The amine functionality facilitates derivatization with fluorophores, chelators, or affinity tags. In coordination chemistry, the ethynyl group of 3-aminophenylacetylene can bind to transition metal centers, forming stable complexes that exhibit interesting catalytic or electronic behavior. Such complexes have been explored in organometallic catalysis, where the acetylene moiety interacts with metals such as palladium, platinum, or ruthenium, and the amino group modulates the electronic environment of the complex. The presence of both amino and ethynyl substituents in a single molecule makes 3-aminophenylacetylene a synthetically flexible and functionally rich intermediate, suitable for the development of new materials, biologically active molecules, and organometallic compounds across multiple scientific disciplines. References 2016. Facile strategy for preparation of alkyne-functionalized cellulose fibers with click reactivity. Cellulose, 24(2). DOI: 10.1007/s10570-016-1153-1 2018. Ribosome-Templated Azide-Alkyne Cycloadditions Using Resistant Bacteria as Reaction Vessels: in Cellulo Click Chemistry. ACS Medicinal Chemistry Letters, 9(9). DOI: 10.1021/acsmedchemlett.8b00248 2024. Synthesis and antitumor activity of dolutegravir derivatives bearing 1,2,3-triazole moieties. BMC Chemistry, 18(1). DOI: 10.1186/s13065-024-01205-3 |
| Market Analysis Reports |