Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Hubei Tuoyuan Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemty.com | |||
 | +86 (027) 8470-0466 | |||
 | +86 (027) 8470-0466 | |||
 | 59741184@qq.com | |||
 | QQ Chat | |||
 | WeChat: 13260668040 | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Puyang Hongda Shengdao New Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hdsdchem.com | |||
 | +86 (393) 489-6585 | |||
 | +86 (393) 489-6585 | |||
 | pyhdshawe@163.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Georganics Ltd. | Slovakia | |||
|---|---|---|---|---|
 | www.georganics.sk | |||
 | +421 (33) 640-3132 | |||
 | +421 (33) 640-3133 | |||
 | georganics@georganics.sk | |||
| Chemical manufacturer since 1998 | ||||
| Classification | Chemical reagent >> Organic reagent >> Cyanate ester / isocyanate |
|---|---|
| Name | 3,5-Dimethylphenyl isocyanate |
| Synonyms | 3,5-Dimethyl-1-isocyanatobenzene; 1-Isocyanato-3,5-dimethylbenzene; 3,5-Xylylisocyanate |
| Molecular Structure | 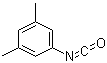 |
| Molecular Formula | C9H9NO |
| Molecular Weight | 147.17 |
| CAS Registry Number | 54132-75-1 |
| EC Number | 258-987-0 |
| SMILES | CC1=CC(=CC(=C1)N=C=O)C |
| Density | 1.0±0.1 g/cm3 Calc.*, 1.045 g/mL (Expl.) |
|---|---|
| Boiling point | 229.9±9.0 °C 760 mmHg (Calc.)*, 205 °C (Expl.) |
| Flash point | 81.1±21.3 °C (Calc.)*, 28.3 °C (Expl.) |
| Index of refraction | 1.516 (Calc.)*, 1.528 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS06;GHS07;GHS08 Danger Details GHS06;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H302-H312-H315-H319-H332-H334-H335 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P233-P260-P261-P264-P264+P265-P270-P271-P280-P284-P301+P316-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P342+P316-P362+P364-P403-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
3,5-Dimethylphenyl isocyanate is an organic compound classified as an aromatic isocyanate, featuring an isocyanate functional group (-N=C=O) attached to a phenyl ring substituted with methyl groups at the 3 and 5 positions. The presence of these methyl groups influences both the chemical reactivity and physical properties of the compound, making it useful in specific synthetic applications. The compound is part of the broader class of aromatic isocyanates, which have been studied and utilized extensively since the early 20th century. Aromatic isocyanates are typically synthesized by reacting the corresponding aromatic amines with phosgene or similar reagents to introduce the reactive isocyanate group. The methyl substitutions at the 3 and 5 positions can modify the electron density of the phenyl ring and affect steric factors, impacting reactivity with nucleophiles such as amines and alcohols. 3,5-Dimethylphenyl isocyanate serves primarily as an intermediate in organic synthesis. Its high reactivity towards nucleophilic species enables the formation of urea, carbamate, and thiourea derivatives. These products find applications in pharmaceuticals, agrochemicals, and polymer industries. In particular, the compound’s reactivity makes it valuable for producing specialty polyurethanes and other polymeric materials with tailored properties. Due to the reactive nature of the isocyanate group, 3,5-dimethylphenyl isocyanate must be handled with care. It is sensitive to moisture and can hydrolyze to form corresponding amines and carbon dioxide, which can affect its stability and storage requirements. It is typically stored under dry, inert atmospheres and protected from humidity to maintain purity. In polymer chemistry, aromatic isocyanates including 3,5-dimethylphenyl isocyanate are integral in the synthesis of polyurethane foams, coatings, and elastomers. While industrial-scale polyurethane production more commonly employs simpler diisocyanates, substituted phenyl isocyanates are used in research and specialty applications to impart unique physical or chemical characteristics to polymer products. In pharmaceutical and agrochemical research, derivatives of substituted phenyl isocyanates are explored for their potential biological activities, including enzyme inhibition and antimicrobial properties. Modifications on the phenyl ring such as methyl groups help tune the compound’s activity and solubility profiles. Safety considerations are important when working with 3,5-dimethylphenyl isocyanate, as isocyanates are known respiratory sensitizers and skin irritants. Appropriate protective measures including ventilation, gloves, and eye protection are essential to minimize exposure risks. In summary, 3,5-dimethylphenyl isocyanate is a reactive aromatic isocyanate used mainly as an intermediate in the synthesis of various organic compounds. Its methyl substitutions influence its reactivity and physical characteristics, supporting applications in pharmaceuticals, agrochemicals, and specialty polymers. Proper handling is critical to ensure safety and maintain compound integrity. References 2000. A study of the thermodynamics and influence of temperature on chiral high-performance liquid chromatographic separations using cellulosetris(3,5-dimethylphenylcarbamate) coated zirconia stationary phases. Chromatographia, 52(9-10). DOI: 10.1007/bf02789747 2023. Optimization of the regioselective synthesis of mixed cellulose 3,5-dimethylphenyl and α-phenylethyl carbamate selectors as separation phases for chiral HPLC. Cellulose, 30(3). DOI: 10.1007/s10570-022-05007-5 2024. Structures of 1,4,2-dithiazolidine 1,1-dioxide and 1,2,4-thiadiazolidin-3-one 1,1-dioxide heterocycles formed from N-benzyl-1-chloromethanesulfonamide and either phenyl isothiocyanate or 3,5-dimethylphenyl isocyanate. Journal of Chemical Crystallography, 54(4). DOI: 10.1007/s10870-024-01034-1 |
| Market Analysis Reports |