Online Database of Chemicals from Around the World
| Shenyang OllyChem Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ollychem.com | |||
 | +86 (24) 6225-9849 +86 13840042106 | |||
 | +86 (24) 6225-9831 | |||
 | info@ollychem.com oliverdu@ollychem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Cayman Chemical Company | USA | |||
|---|---|---|---|---|
 | www.caymanchem.com | |||
 | +1 (734) 971-3335 | |||
 | +1 (734) 971-3640 | |||
 | sales@caymanchem.com | |||
| Chemical manufacturer | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Hormone and endocrine-regulating drugs >> Prostaglandins |
|---|---|
| Name | (+)-Cloprostenol |
| Synonyms | (5Z)-7-[(1R,2R,3R,5S)-2-[(1E,3R)-4-(3-Chlorophenoxy)-3-hydroxy-1-buten-1-yl]-3,5-dihydroxycyclopentyl]-5-heptenoic acid |
| Molecular Structure | 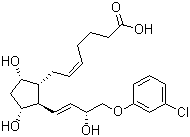 |
| Molecular Formula | C22H29ClO6 |
| Molecular Weight | 424.92 |
| CAS Registry Number | 54276-21-0 |
| EC Number | 682-029-4 |
| SMILES | C1[C@@H]([C@@H]([C@H]([C@@H]1O)/C=C/[C@H](COC2=CC(=CC=C2)Cl)O)C/C=CCCCC(=O)O)O |
| Density | 1.3±0.1 g/cm3, Calc.*, 1.321 |
|---|---|
| Index of Refraction | 1.623, Calc.* |
| Boiling Point | 628.0±55.0 °C (760 mmHg), Calc.* |
| Flash Point | 333.6±31.5 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H317-H334-H360 Details | ||||||||
| Safety Statements | P203-P233-P260-P261-P264-P270-P271-P272-P280-P284-P301+P317-P302+P352-P304+P340-P318-P321-P330-P333+P317-P342+P316-P362+P364-P403-P405-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
|
(+)-Cloprostenol is a synthetic analog of prostaglandin F2α and belongs to the class of prostaglandin derivatives, which are known for their potent biological activities. It was first developed in the late 20th century as a veterinary medication with applications in reproductive health, particularly in livestock. The compound's structure features a cyclopentane ring and several functional groups that contribute to its biological activity, making it an important tool in animal husbandry. The discovery of (+)-cloprostenol was driven by the need to improve reproductive management in agricultural practices. The compound is recognized for its ability to induce luteolysis, which is the regression of the corpus luteum, leading to a decrease in progesterone levels. This action facilitates the synchronization of estrus cycles in livestock, allowing for better timing of breeding and improved reproductive efficiency. The ability to control reproductive cycles is crucial for maximizing productivity in the farming industry, particularly in cattle, sheep, and pigs. In veterinary medicine, (+)-cloprostenol is utilized for various reproductive purposes, including the induction of estrus, treatment of cystic ovarian disease, and the termination of pregnancies. Its effectiveness in managing reproductive health has made it a valuable agent in the field of animal agriculture, where precise control over breeding times is essential for optimizing herd productivity. The pharmacological properties of (+)-cloprostenol extend beyond reproductive applications. Research has also explored its potential use in human medicine, particularly in managing conditions like glaucoma and promoting labor in obstetric practices. The compound’s mechanism of action involves binding to specific prostaglandin receptors, which mediates its biological effects. Moreover, (+)-cloprostenol has been studied for its anti-inflammatory properties and potential use in treating conditions that involve aberrant inflammatory responses. The compound's influence on smooth muscle contraction and vasodilation has also led to investigations into its cardiovascular effects, although most applications remain focused on reproductive health. In conclusion, (+)-cloprostenol is a significant synthetic prostaglandin analog that has transformed reproductive management in veterinary medicine. Its discovery has led to enhanced productivity in livestock and improved reproductive outcomes, highlighting the importance of chemical compounds in agricultural practices. Continued research may reveal further therapeutic potentials, expanding the applications of this versatile compound beyond veterinary use. References 2023. Some aspects of the synthesis and modification of cross-conjugated cyclopentenone prostaglandins. Russian Chemical Bulletin. DOI: 10.1007/s11172-023-4026-4 2003. Update on prostaglandin analogs. Current Opinion in Ophthalmology. DOI: 10.1097/00055735-200304000-00001 1979. Veterinary Uses of Prostaglandins. Practical Applications of Prostaglandins and their Synthesis Inhibitors. DOI: 10.1007/978-94-011-7843-3_10 |
| Market Analysis Reports |