Online Database of Chemicals from Around the World
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Taizhou KEDE Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kedechemical.com | |||
 | +86 (576) 8461-3060 +86 13093829633 | |||
 | +86 (576) 8461-3060 | |||
 | sales@kedechemical.com | |||
 | QQ Chat | |||
 | WeChat: 13093829633 | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Shanghai Forever Synthesis Co.,Ltd. | China | |||
|---|---|---|---|---|
 | www.foreversyn.com | |||
 | +86 (551) 6288-8437 +86 18096409024 | |||
 | sales@foreversyn.com sales02@foreversyn.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Classification | Organic raw materials >> Alcohols, phenols, phenolic compounds and derivatives |
|---|---|
| Name | (+)-5-trans-Cloprostenol |
| Synonyms | [1R-[1alpha(E),2beta(1E,3R*),3alpha,5alpha]]-7-[2-[4-(3-Chlorophenoxy)-3-hydroxy-1-butenyl]-3,5-dihydroxycyclopentyl]-5-Heptenoic acid |
| Molecular Structure | 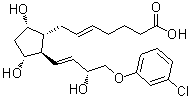 |
| Molecular Formula | C22H29ClO6 |
| Molecular Weight | 424.91 |
| CAS Registry Number | 57968-81-7 |
| EC Number | 832-856-3 |
| SMILES | C1[C@@H]([C@@H]([C@H]([C@@H]1O)/C=C/[C@H](COC2=CC(=CC=C2)Cl)O)C/C=C/CCCC(=O)O)O |
| Density | 1.3±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 628.0±55.0 °C 760 mmHg (Calc.)* |
| Flash point | 333.6±31.5 °C (Calc.)* |
| Solubility | Practically insoluble (0.032 g/L) (25 °C), Calc. |
| Index of refraction | 1.623 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302 Details | ||||||||
| Safety Statements | P264-P270-P301+P317-P330-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
|
(+)-5-trans-Cloprostenol is a stereoisomeric derivative of Cloprostenol, which is a synthetic analogue of Prostaglandin F2α (PGF2α). Its molecular formula is C22H29ClO6 with a molecular weight of approximately 424.9. The compound retains the cyclopentane core and the heptenoic acid side chain characteristic of prostaglandin F analogues, but features a trans configuration at the 5-position double bond in the omega-side chain. The “(+)” designation indicates its optical activity, corresponding to a specific enantiomer. Cloprostenol and its stereoisomers act primarily as agonists at the FP prostanoid receptor. Activation of these receptors induces luteolysis, promotes uterine contraction, and modulates estrus in livestock. The (+)-5-trans isomer exhibits significantly lower biological activity than the 5-cis isomer, which is the primary therapeutically active form. In animal models, the trans isomer is reported to be several times less potent than the cis form for reproductive applications such as estrus induction and termination of pregnancy. The synthesis of (+)-5-trans-Cloprostenol occurs as a minor stereoisomeric product during the preparation of Cloprostenol. The precise stereochemistry of the double bond and hydroxyl substituents on the cyclopentyl ring influences receptor binding and pharmacological potency. The presence of the 3-chlorophenoxy moiety and hydroxyl groups ensures selectivity for the FP receptor while enhancing chemical stability relative to the free prostaglandin. Applications of (+)-5-trans-Cloprostenol are largely limited to research, analytical characterization, and stereochemical studies. Unlike the cis isomer, it does not have independent regulatory approval or clinical usage in veterinary medicine due to its reduced potency. However, its identification and characterization are important for quality control and understanding stereochemistry-activity relationships in prostaglandin analogues. Overall, (+)-5-trans-Cloprostenol represents a stereoisomer of Cloprostenol with defined optical and geometric properties, reduced biological activity, and value primarily in research and analytical contexts rather than direct therapeutic use. References Sharif NA, Kelly CR, Crider JY (2002) Agonist activity of prostaglandin analogs at the cloned human ciliary body FP prostaglandin receptor. Journal of Ocular Pharmacology and Therapeutics 18 313–324 DOI: 10.1089/10807680260218489 Woodward DF, Krauss AH, Chen J, Liang Y (2008) Pharmacological characterization of prostaglandin analogs used in glaucoma therapy: prostamide-related activity of bimatoprost. Journal of Ocular Pharmacology and Therapeutics 24 475–482 DOI: 10.1124/jpet.102.047837 |
| Market Analysis Reports |